Disclosures for Andrew D. Zelenetz, MD, PhD
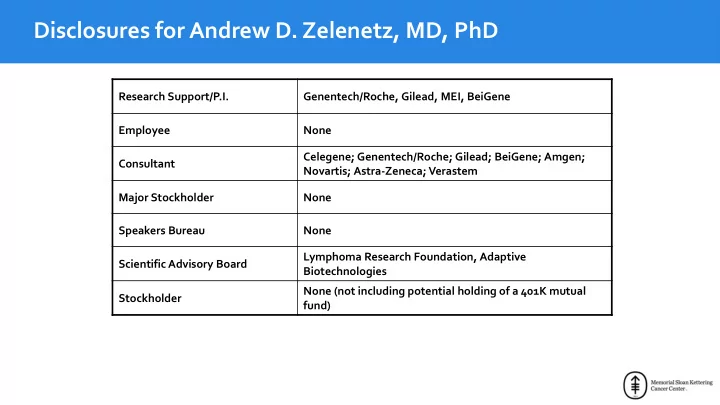
Research Support/P.I. Genentech/Roche, Gilead, MEI, BeiGene Employee None Consultant Celegene; Genentech/Roche; Gilead; BeiGene; Amgen; Novartis; Astra-Zeneca; Verastem Major Stockholder None Speakers Bureau None Scientific Advisory Board Lymphoma Research Foundation, Adaptive Biotechnologies Stockholder None (not including potential holding of a 401K mutual fund)