SLIDE 1
anti-Bromination of alkenes: direct bromonium ion intermediate - - PowerPoint PPT Presentation
anti-Bromination of alkenes: direct bromonium ion intermediate - - PowerPoint PPT Presentation
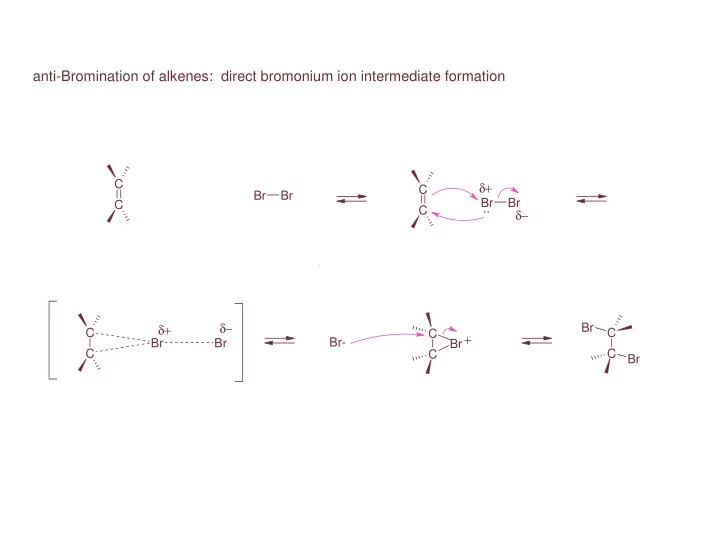
anti-Bromination of alkenes: direct bromonium ion intermediate formation C + C Br Br Br Br C .. C + Br C C C + Br Br Br- Br C C C Br Orbital interactions in the anti-Bromination of alkenes: direct
SLIDE 2
SLIDE 3
Alkyl substituents on the bromomium ion desymmetrize it: the more substitued carbon can bear more
- f the burden of the bromine’s positive charge:
So, the electrophilic Br ends up on the less substituted carbon and the nucleophilic Br ends up on the more substituted carbon.
C C H H Br Br C C H H Br Br C C H H Br Br C C Br H H C C H H Br Br δ+ δ− Br- .. δ+ δ− δ+ δ+
SLIDE 4
This is a Markovnikov-like addition: when adding HCl, the electrophile (H) ends up on the less substituted carbon and the nucleophile (Cl) ends up on the more substituted carbon. Although the mechanism is different, the regiochemistry is not: C C H H H Cl C C H H H C C H H H Cl Cl- δ+ δ− +
SLIDE 5
H H H Cl H Cl Cl H Cl H H H Cl Cl Cl H Cl H H Cl Cl H Cl H H Cl Cl2 CH2Cl2 H H H H Cl Cl Cl H Cl H Cl H Cl H H Cl H Cl Cl2 CH2Cl2
Anti-addition of Cl2 to trans-3-hexene meso Anti-addition of Cl2 to cis-3-hexene enantiomers
SLIDE 6
R H H H R O H O H H
- 1. O3
- 2. Zn, H2O
+ R H R
2
H R O H O R
2
H
- 1. O3
- 2. Zn, H2O
+ R R
1
H H R O R
1
O H H
- 1. O3
- 2. Zn, H2O
+ R R
1
R
2
H R O R
1
O R
2
H
- 1. O3
- 2. Zn, H2O
+ R R
1
R
2
R
3
R O R
1
O R
2
R
3
- 1. O3
- 2. Zn, H2O
+
Ozonolysis of alkenes
aldehyde ketone mono formaldehyde ketone tri aldehyde terminal di formaldehyde ketone aldehyde tetra ketone internal di (E) or (Z) aldehyde
SLIDE 7
H O H H O
- 1. O3
- 2. Zn, H2O
It doesn’t matter if the alkene is a cycloalkene:
SLIDE 8
Addition reactions of carbon-carbon double bonds Reaction Adds Regiochemistry Diastereoselective? Rearrangement observed? Hydrohalogenation H–X Markovnikov No Yes H–Br AntiMarkovnikov No No Hydration Acid-catalyzed Hydration H–OH Markonikov No Yes Oxymercuration- demercuration H–OH Markovnikov No No Borohydration-
- xidation
H–OH AntiMarkovnikov Yes - syn No Hydrogenation H–H
- Yes - syn
No * Halogenation X–X
- Yes - anti
No Halohydroxylation X–OH Markovnikov Yes - anti No Dihydroxylation Epoxidation- acid hydroylsis HO–OH
- Yes - anti
No * OsO4 HO–OH
- Yes - syn
No Oxidative cleavage O + O
- No