HOME
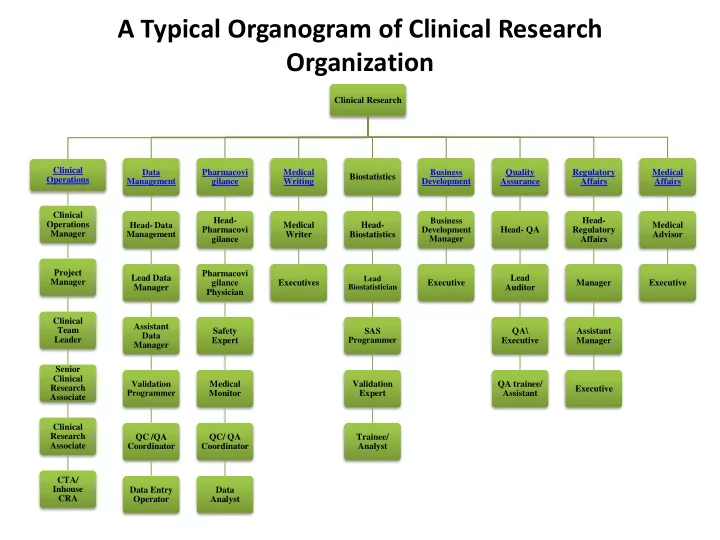
A Typical Organogram of Clinical Research Organization
Clinical Research Clinical Operations Clinical Operations Manager Project Manager Clinical Team Leader Senior Clinical Research Associate Clinical Research Associate CTA/ Inhouse CRA Data Management Head- Data Management Lead Data Manager Assistant Data Manager Validation Programmer QC /QA Coordinator Data Entry Operator Pharmacovi gilance Head- Pharmacovi gilance Pharmacovi gilance Physician Safety Expert Medical Monitor QC/ QA Coordinator Data Analyst Medical Writing Medical Writer Executives Biostatistics Head- Biostatistics
Lead Biostatistician
SAS Programmer Validation Expert Trainee/ Analyst Business Development Business Development Manager Executive Quality Assurance Head- QA Lead Auditor QA\ Executive QA trainee/ Assistant Regulatory Affairs Head- Regulatory Affairs Manager Assistant Manager Executive Medical Affairs Medical Advisor Executive