Pure Component 2 Pure Component 1 Mixture 12
in
W
- ut
Q
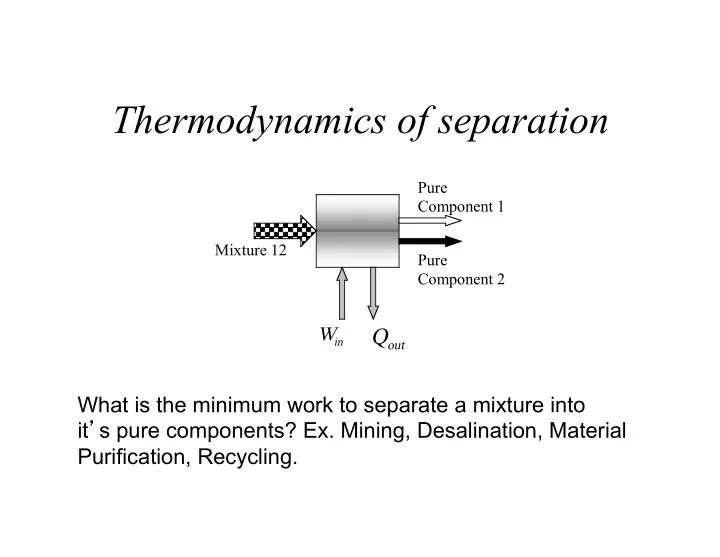
Thermodynamics of separation
What is the minimum work to separate a mixture into it’s pure components? Ex. Mining, Desalination, Material Purification, Recycling.
W in = ! N 12 ( h mix ! T 0 s mix ) + T 0 S irr Minimum Work of - - PowerPoint PPT Presentation
Thermodynamics of separation Pure Component 1 Mixture 12 Pure Component 2 W Q in out What is the minimum work to separate a mixture into it s pure components? Ex. Mining, Desalination, Material Purification, Recycling. Balance Eq ns
Pure Component 2 Pure Component 1 Mixture 12
in
W
Q
What is the minimum work to separate a mixture into it’s pure components? Ex. Mining, Desalination, Material Purification, Recycling.
Pure Component 2 Pure Component 1 Mixture 12
in
W
Q
dNi,sys dt = Ni,in ! Ni,out
dE dt = ! Qout + Win + H12 ! H1 ! H2
dS dt = ! Qout T0 + S12 ! S1 ! S2 + Sirr
Sirr
mix + T0
mix
mix = Δho mix –T0 Δso mix.
mix ≈ –T0 Δsmix =
mix < 0
* at standard conditions
How many ways can “r” atoms be positioned in a lattice with “n” locations?
i=1 j
)) )) x ln( N x ln ln N ( R T W
) N ( min min
i
! + ! = 1
2 1
)) 1 ln( ln ) 1 ((
2 1 ) 1 ( min
1
x N x N R T W N ! + ! ! =
!
ex,O2
xO2 ) ! 298(K) # 8.314(J / molK)ln(0.212) = 3.84(kJ / mol)
In wet air you get 3.97 kJ/mol : compare with Szargut
Table from the EngineeringToolBox.com
Energy kg(target) = kg (processed) kg (target) i Energy kg (processed) ~ 1 g i Energy kg (processed)
Chapman and Roberts p 113 & 116 underground ~ 1000/g (MJ/t metal)
Sherwood plot showing the relationship between the concentration
the target material [Grübler 1998].
x x, mixture =
x, i + RT
CRUST at To, po Ore value at mine Pure ore (e.g. Fe2O3) Pure metal Metal alloy Mixing in product Mixing in waste stream Further mixing and corrosion Exergy Purification Stages Recycle to pure metal
Theoretical Exergy Values for a metal extracted from the earth’s crust shown at various stages of a product life cycle (not to scale)