SLIDE 1
Slide 1
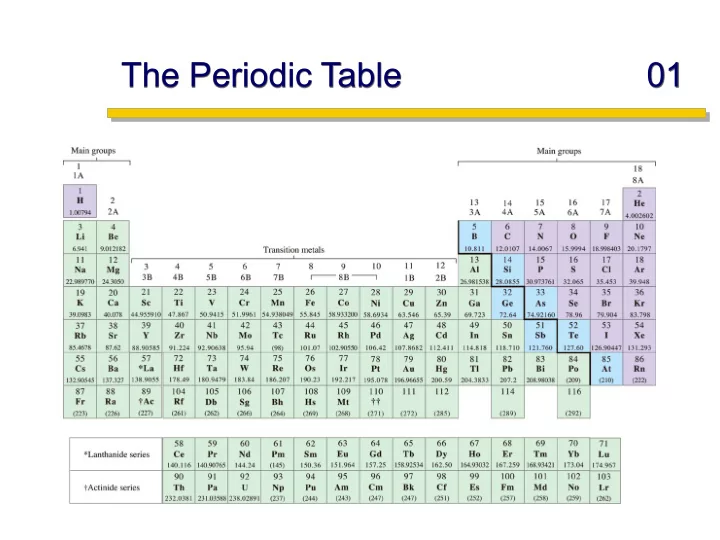
The Periodic Table 01 Slide 1 Electron Radial Distribution 03 p - - PowerPoint PPT Presentation
The Periodic Table 01 Slide 1 Electron Radial Distribution 03 p Orbital Shapes: http://www.shef.ac.uk/chemistry/orbitron/ Slide 2 Electron Radial Distribution 04 d and f Orbital Shapes: Slide 3 Shapes of Orbitals 01 Give a
Slide 1
Slide 2
Slide 3
Slide 4
Slide 5
Slide 6
Slide 7
Slide 8
Slide 9
Slide 10
Slide 11
Slide 12