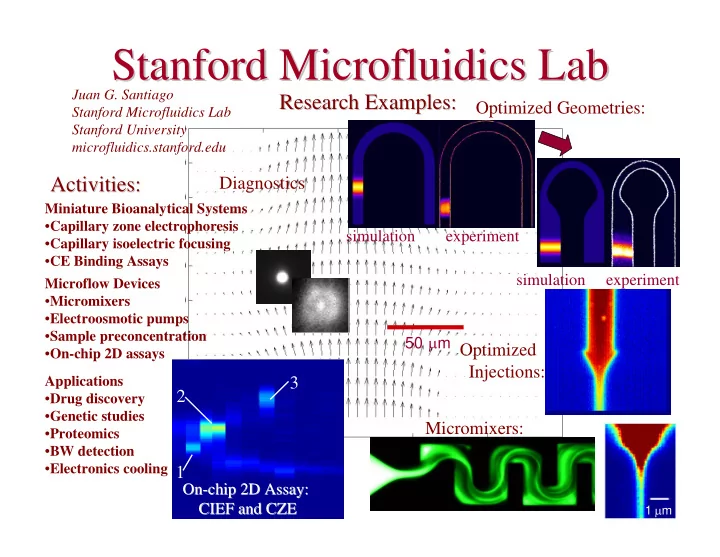
SLIDE 21 1998
Highest ITP stacking Run-time: 6-10 min L = 53.5 cm
5500
LE: 10 mM NaOH titrated with H3PO4, TE: 6.13 mM THeACl titrated with H3PO4 NXX-066
Capillary UV
ITP
2000
Run-time: 10-13 min L = 64 cm
200
50 mM phosphoric acid with 20 % acetonitrile, DI water 1-naphthylamine, laudanosine
Capillary UV
FASI
1999
Run-time: 15-17 min L = 25 cm
1000
75 mM phosphate, DI water Bromide, nitrate, bromate
Capillary UV
FASI
1999
Run-time: 5-10 min L = 61 cm
300
1 mM phosphoric acid, 40 mM potassium dihydrogen phosphate Maleic, fumaric acids, bromide, nitrate
Capillary UV
LVSS
1996
Run-time: 4-6 min L = 24.6 cm
1000
45 mM NaH2PO4 and 15 mM Na2HPO4, 60% v/v 1-propanol Dese, Amino
Capillary UV
FASS
1992
First LVSS Run-time: 6-10 min L d) = 65 cm
histidine, DI water PTH-aspartic acid, PTH-glutamine acid
Capillary UV
LVSS
2005
Run-time: 2 min
500
LE: 250 mM NaCl, TE: 95 mM TAPS, 73 mM TEA Fluorescein
Microchip Fluore- scence
ITP
2002
Run-time: 1-2 min
530
LE: 25 mM imidazole, 20 mM HCl, TE: 160 mM imidazole, 40 mM HEPES eTags
Microchip Fluore- scence
ITP
2003
Narrow sample channel Run-time: 2 min
80
175 mM Phosphate, DI water Fluorescein disodium salt
Microchip Fluore- scence
FASS
2003
Five-channel geometry. Run-time: 1 min.
100
HEPES (0.1 mM and 100 mM), NaCl (0.2 mM and 200 mM) Fluorescein sodium salt
Microchip Fluore- scence
FASS
2001
Six-channel geometry Run-time: 2 min
65
Carbonate (200 μM and 32 mM) FITC-arginine
Microchip Fluore- scence
FASS
1995
First on-chip FASS Run-time: 20 sec
13.8
Sodium tetraborate (0.5 mM and 500 mM) didansyl-lysine
Microchip Fluore- scence
FASS
Ref. Comments SE Electrolyte Sample Microchip/ capillary Detection Mode Method