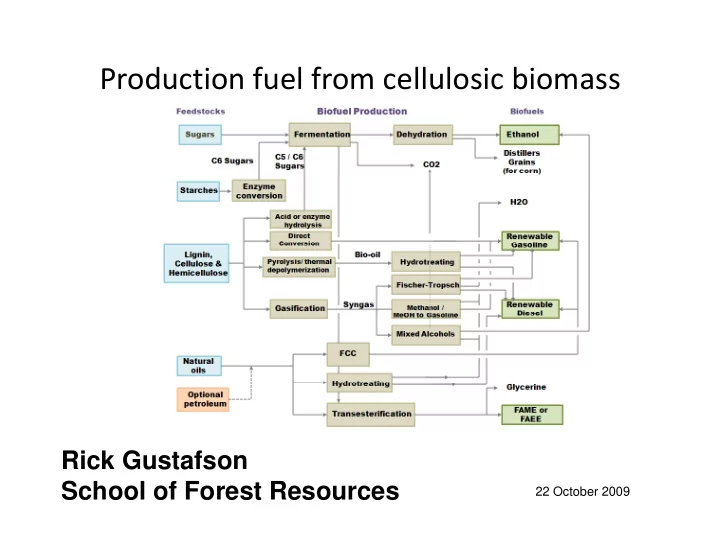
Production fuel from cellulosic biomass
Ri k G t f Rick Gustafson School of Forest Resources
22 October 2009
Production fuel from cellulosic biomass Ri k G Rick Gustafson t f - - PowerPoint PPT Presentation
Production fuel from cellulosic biomass Ri k G Rick Gustafson t f School of Forest Resources 22 October 2009 Motivation for biofuels Motivation for biofuels Climate Change Biomass is renewable and can be carbon neutral Corn ethanol
22 October 2009
Source: National Geographic
http://www.nationmaster.com/red/graph/ene_oil_exp_net-energy-oil-exports-net&b_map=1#
‘Peak Oil’ Is a Waste of Energy – NY Times 24 August 2009
BP Finds Giant Oil Field Deep in G lf f M i Gulf of Mexico NY Times 2 Sept. 2009
Fires 33.5 M mt/2006
Figure 4 4 2 Washington historic CO2e emissions by sector with fire emissions superimposed (adapted from Waterman-Hoey and Figure 4.4.2. Washington historic CO2e emissions by sector with fire emissions superimposed (adapted from Waterman-Hoey and Nothstein 2007, Wiedinmyer and Neff 2007).
WA GHG Reduction Goals 2020 to 1990 2050 to 50% 1990 2035 to 75% 1990 2050 to 50% 1990
Waterman‐Hoey and Nothstein. 2007. Washington’s Greenhouse Gas Emissions: Sources and Trends ‐ 2006. WA Dept. of Community, Trade and Economic Development. Olympia, WA. 18 pp. http://www.cted.wa.gov/site/853/default.aspx
Current ethanol capacity = 10.6 billion gal/year Current biodiesel capacity = 700 million gal/year
Source: http://www.ethanolrfa.org/industry/statistics,
Ethanol futures, $/gal
40 30 35 Year)
2005 EPAct RFS Current Ethanol Production 2007 RFS - Conventional 2007 RFS - Cellulosic Biofuels 2007 RFS - Biomass-based Diesel 2007 RFS - Unaffiliated
20 25 Billion Gallons per
Cellulosic
10 15 Renewable Fuel (B mated 5 R Estim
Corn
2000 2005 2010 2015 2020
Total US oil consumption = 320 billion gals/year Total US oil imports = 220 billion gals/year
U d t d t A il 2005
13
The “Billion Ton” potential represents about 1/3 of our petroleum use when The Billion Ton potential represents about 1/3 of our petroleum use when converted to liquid fuels Billion Barrel of Oil Equivalents
Based on ORNL & USDA Resource Assessment Study by Perlach et.al. (April 2005) http://www.eere.energy.gov/biomass/pdfs/final_billionton_vision_report2.pdf
Source: WSU, WA DOE. http://www.ecy.wa.gov/pubs/0507047.pdf
Fuels Ethanol Renewable Diesel Mi d Al h l
Mixed Alcohols Dimethyl Ether Power Electricity Heat Chemicals Plastics Solvents Chemical Intermediates Phenolics
Enzymatic Fermentation Gas/liquid Fermentation Trees Grasses
Adhesives Furfural Fatty Acids Acetic Acid Carbon Black Et
Acid Hydrolysis/ Fermentation Gasification Pyrolysis Agricultural Crops Residues Animal Wastes M i i l S lid W t
10/29/2009 17
Etc. Food and Feed
Pyrolysis Combustion Co-firing Municipal Solid Waste
Adapted from Holmgren etal (2008); Hydrocarbon Processing, Sep.
Cellobiose Unit
O O O O O O O O
CH2OH OH HO OH HO CH2OH OH CH2OH HO OH CH2OH HO O β β β β
2
β β
Cellulose
5 ca bo suga s: y ose, a ab ose
O O O O O
OH HO HO OH
O O O O
OH HO
O O
HO OH
O O CO2H H3CO OH HOH2C O O CO2H OH
OH OCH3 CH3 CH CH H2COH 16 H2COH CH2 CH2 H C 26 CH 28 27 O CH2OH H CH3 CH O O H2COH H3CO OCH3 COH H HC CH2OH OH CH2OH C O H3CO CH 6 OCH3 O O C CH O H3C H CH3 CH OH O H3C 15 16 Carbohydrate CH2OH OH OCH3 HC 14 O C O OCH3 24 25 H2COH H H3CO CH CH O O C O CH H C H C HOH2 HO OCH3 OH 4 5 7 H HC CH CH2 O C CH O H2C 8 HO CH2OH H3C 13 O CH O CH O H2COH HC CH O 24 H2COH O HC H 23 COH OCH CH HO H CH C O H2C CH O O CH2OH H3C 2 3 O O CH H3C 8 OHC CH CH2OH CH2OH O O H3C 9 10 H3C 12 CHO 17 HO CH O O H C O O CH CH O COH H2 C H H2COH OCH3 C CH CH CHO 22 OCH3 C O O O CH H3C CH2OH 1 C OH 10 O HC CH COH H2 CH2 O C H 11 O C O CH2 H3C 18 H CH O O H H3C 19 COH H COH H2 H O HC O C OCH3 21 C H2 O COH H HCOH O OH H3C OH OCH3 20
Feedstock Glucan (cellulose) (%) Xylan (hemicellulose) (%) Lignin (%) Corn stover 37.5 22.4 17.6 Corn fiber 14 3 16 8 8 4 Corn fiber 14.3 16.8 8.4 Pine wood 46.4 8.8 29.4 Poplar 49.9 17.4 18.1 p Wheat straw 38.2 21.2 23.4 Switchgrass 31.0 20.4 17.6 g Chemical composition of biomass (adapted from Mosier et al., 2005).
Ethanol Ethanol
J.A. Dumesic / Catalysis Today 111 (2006) 119–132
Schematic of goals of pretreatment on lignocellulosic material (adapted from Hsu et al., 1980).
Increases accessible surface area Decrystalizes cellulose Removes hemicellulose Removes lignin Alters lignin structure surface area
Uncatalyzed steam explosion ■ ■ Liquid hot water ■ ND ■ q pH controlled hot water ■ ND ■ ND Flow-through liquid ■ ND ■ hot water ■ ND ■ Dilute acid ■ ■ ■ Flow-through acid ■ ■ ■ AFEX ■ ■ ■ ■ ARP ■ ■ ■ ■ Lime ■ ND ■ ■ Adapted from Mosier et al., 2005
Minor effect Major effect
E do beta 1 4 lu a a e a t ithi the hai b eaki it » Endo‐beta‐1,4‐glucanase acts within the chain, breaking it into smaller units and providing more ʺendsʺ for CBH. » Cellobiohydrolase (CBH), acts on the end of the molecule successively cleaving off the disaccharide cellobiose successively cleaving off the disaccharide cellobiose. » Beta‐glucosidase (or cellobiase) which cleaves cellobiose to two glucose units.
Binding domain Catalytic domain Binding domain Catalytic domain
E d l
(EG)
Endoglucanases (EG)
cutting the cellulose chains randomly
Cellobiohydrolyses (CBH) cutting cellobiose units of the ends of the cellulose chains
Cellular metabolism under anaerobic conditions for the production of energy and metabolic intermediates
» Butanol » Acetic acid » Acetic acid » Propionic acid » Lactic acid
processes producing liquids as the primary product p p g q p y p
reducing conditions to produce primarily permanent gases, with char, water, and condensibles as minor products
10/29/2009 32
Biomass Conversion to Fuels – Major Biochemical Conversion Steps Major Biochemical Conversion Steps
Feed Processing and Handling
Gasification Pyrolysis Gas Cleanup High T Separation Gas Conditioning Collection/Fractionation Fuel Synthesis Upgrading Heat & Power y y Separation pg g
O bl
– FT liquids – MTG
Benzene removal
H2/CO adjustment
MTG – Mixed OH
10/29/2009 33
Th di t ib ti f d t d d t t d id ti
Liquid Char Gas
The distribution of products depends on temperature and residence time
FAST PYROLYSIS 75% 12% 13% moderate temperature short residence time short residence time GASIFICATION 5% 10% 85% high temperature long residence time
34 Source: Bridgewater and Czernik
Primary Energy Source Syngas Step Conversion Technology Products
Fischer Tropsch (FT) Upgrading Naphtha Diesel
Syngas to Liquids (GTL) Process
Natural Gas
(FT) Lubes
Syngas to Chemicals Technologies
Coal Syngas (CO + H2)
Syngas to Chemicals Technologies
M th l Acetic Acid Methanol
Biomass
Hydrogen Mixed Alcohols (e.g. ethanol, propanol) Others (e.g. Triptane, DME, etc)
Extra Heavy Oil
10/29/2009 35 Source: BP
absence of oxygen absence of oxygen
gasification processes
charcoal and chemicals for thousands years
10/29/2009 36
Biocrude is water miscible and is comprised of many oxygenated
Combustible
D it 1 2 k /l
“ ”
10/29/2009 37 Source: Bridgewater and Czernik
10/29/2009 38
Source: Bridgewater and Czernik
Forest
Upgrade biooil at refinery
Forest Residuals BioOil Pyrolysis
Much cheaper to ship biooil than biomass beca se of higher energ
39
FOREST biomass because of higher energy density
Cellulosic ( C) Cellulosic ( C) Cellulosic ( C) Ethanol Corn Ethanol S Pyrolysis O Methanol Ethanol (BC) Ethanol (TC) Steam Ethanol (TC) POX Dry Mill Sugar Cane Fuel Oil Intermediate Intermediate
Yield
gal/ton biomass
90 80 77 102 12.3 137 149
gal Ethanol eq/ton biomass
90 80 77 102 12.3 135 115 B P d t By-Product
type
C3+OH DDGS Sugar
15 33 wt% 5.7 wt%
196
Nat Gas, MMBtu/dry ton
3.11 Process Efficency, HHV
Overall (Prod + Byprod) %
48 47 46 74 68 55
Product %
44 40 37 44 67 55 Carbon Utilization (basis feed carbon) Carbon Utilization (basis - feed carbon)
Product %
31 27 26 39 59 35
By-Product %
5 37
16 15 24 19
Combustion Flue Gas CO2 %
53 52 36 5 41 47
Other % Type
char NG Flue Gas
%
8 10 18
100 MMGPY EtOH Production, $2005$
10/29/2009 41
Geographic, Feedstock, and Technology Diversity
RSE Pulp & Chemical, LLC Bi h i l DOE Great Lakes NewPage Thermochemical Mascoma Biochemical Various (Lebanon, NH) Cargill Inc Biochemical Various (Minneapolis, MN) Regents of the University of Minnesota Various (Minneapolis, MN) Cornell University (I h NY) Biochemical Woody Biomass- Mill Residues (Old Town, ME) Mascoma Biochemical Hardwoods (Upper Peninsula, MI) Flambeau River Thermochemical Forest Residues/Wood Waste (Park Falls, WI) Pacific Ethanol Biochemical Wheat Straw, Stover, Poplar Residues (Boardman, OR) Montana State University Bozeman, (MT) University of Maine (Orono, ME) University of M h A h Poet Biochemical Corn Cob/Corn Fiber (Emmetsburg, IA) DOE Joint Bioenergy Institute (Berkeley, CA) DOE Great Lakes Bioenergy Research Center (Madison, WI) Thermochemical Woody Biomass – Mill Residues (Wisconsin Rapids, WI) DSM Innovation Center Biochemical Various (Parsippany, NJ) Novozymes Biochemical Various Purdue University (West Lafayette, IN) (Ithaca, NY) GE Global Research (Niskayuna, NY) Emery Energy Gas Technology Institute (Des Plaines, IL) UOP, LLC Thermochemical (Des Plaines, IL) Stevens Institute of Technology (Hoboken NJ) Univeristy of Toledo (Toldeo, OH) Massachusetts – Amhers (Amherst, MA) BlueFire Ethanol Biochemical Municipal Solid Waste (M CA) Lignol Biochemical Woody Biomass- Ag Residues (Grand Junction, CO) ICM Biochemical Switchgrass, Forage Sorghum, Stover (St. Joseph, MO) Abengoa Biochemica Agricultural Residue (Hugoton, KS) DOE Bioenergy Science Center (Oak Ridge, TN) (Davis, CA) Genencor Biochemical Various (Palo Alto, CA) Dupont Biochemical Various (Wilmington, DE) Purdue University Biochemical (West Lafayette, IN) Ceres, Inc Various (Thousand Oaks CA) Alltech Biochemical Corn Cobs/Corn Fiber (Washington County, KY) Emery Energy Thermochemical Corn Stover (Salt Lake City, UT) Iowa State (3) University (Ames, IA) Research Triangle Institute (2) Thermochemical Woody Biomass (Research Triangle Park, NC) University of Georgia (Ath GA) ( , ) Virginia Tech (Blacksburg, VA) (Mecca, CA) Range Fuels Thermochemical Woody Waste (Soperton, GA) Verenium Corp Biochemical Various (San Diego, CA)
Nine Small-Scale Biorefinery Projects
(Thousand Oaks, CA) Southern Research Institute Thermochemical Various (Birmingham, AL) (Athens, GA) Georgia Tech (Atlanta, GA)
Regional Partnerships
Four Commercial-Scale Biorefinery Projects Four Improved Enzyme Projects Five Projects for Fermentation Organisms Five Thermochemical Syngas Projects Th Offi f S i Bi C t
Verenium Biofuels Corp. Biochemical Process Energy Cane and Bagasse (Jennings, LA)
g p
South Dakota State Univ., Brookings, SD Cornell University, Ithaca, NY
Oklahoma State Univ., Stillwater, OK Oregon State Univ., Corvallis, OR Three Office of Science Bioenergy Centers DOE Joint Solicitation Biomass Projects Five Thermochemical Bio-Oil Projects Six University Projects Modified 10/1/2008