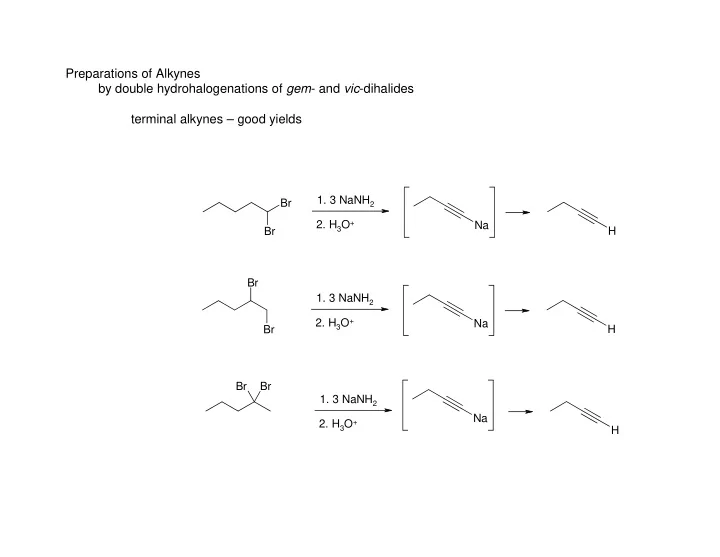
SLIDE 1 Preparations of Alkynes by double hydrohalogenations of gem- and vic-dihalides terminal alkynes – good yields
Br Br H Na Br Br H Na Br Br H Na
- 1. 3 NaNH2
- 2. H3O+
- 1. 3 NaNH2
- 2. H3O+
- 1. 3 NaNH2
- 2. H3O+

SLIDE 2 Br Br H H H H H C H H H N H H C H H C H H H N H H H
+ -NH2 + ..
. .
+ -NH2 .. - + NH3
Preparations of Alkynes by double hydrohalogenations of gem- and vic-dihalides internal alkynes – usually lower yields than terminal alkynes, due to allene by-products and alkyne isomerization; alternative bases mitigate this

SLIDE 3
Preparations of Alkynes internal alkynes because of the above problem, internal alkynes usually are prepared not by elimination but by substitution:
H Br NaNH2 .. -

SLIDE 4
Naturally occurring alkynes
(+)-Laurencin isolated from red algae Laurencia glandulifera Burton, J.W.; Clark, J.S.; Derrer, S.; Stork, TC.; Bendall, J.G.; Holmes, AB.; J. Am. Chem. Soc. 1997, 119, 7483.

SLIDE 5
Thiarubrine A antiviral antifungal (Candida albicans) antibacterial Masato Koreeda, M.; Yang, W. J. Am. Chem. Soc. 1994, 116, 10793.

SLIDE 6 Enediynes
- Share a common (Z)-1,5-diyn-3-ene moiety
- Target minor groove or AT-rich DNA & intercalate
- “Triggered” by an extended conjugate addition
- Result of pulling trigger is a diradical
- Diradical leads to SS and DS DNA cuts
NCZ diradical
Nicolaou, K.C.; Dai, W.-M. Angew. Chem. Int. Ed. Engl. 1991, 30, 1387-1530.

SLIDE 7
Synthesize 2-heptyne from acetylene (ethyne) and any haloalkanes you need

SLIDE 8 Retrosynthesis:
Br H C7 ..
C4 C3

SLIDE 9
Now do the same thing for 1-propyne:

SLIDE 10 So the overall forward synthesis is a 2C + 1C + 4C coupling strategy:
H H C H H CH3 C CH3 Br CH3 NaNH2 THF
THF CH3Br NaNH2 THF
THF

SLIDE 11
Now, provide a multistep synthesis of meso-5,6-decanediol from # 4C sources of carbon and any other reagents you require.
CH2CH2CH2CH3 H O H H O H CH2CH2CH2CH3

SLIDE 12
Is a C10 vicinal diol. ˆ synthesis will involve chain elongation of an alkyne. Diols are made from alkenes. Which alkene?
(Z)-5-decene.
How is (Z)-5-decene made? From an alkyne: 5-decyne
CH2CH2CH2CH3 H O H H O H CH2CH2CH2CH3 H O H H O H

SLIDE 13
Finally, how is this alkyne created from pieces of 4 carbons or fewer? So this is a 2C + 4C + 4C = 10C assembly strategy.
Br Br + +

SLIDE 14
The forward synthesis of meso-5,6-decanediol is: . . .How would a synthesis of (SR, SR)-5,6-butanediol differ from the one just proposed?

SLIDE 15
Synthesize heptanal from acetylene, # 5C compounds, and any other reagents required:
acetylene
heptanal One way: a 2C + 5C = 7C retrosynthetic strategy
H O H H ?
H O H O H H H Br H H is the keto form of the enol: +

SLIDE 16 Forward synthesis:
H O H H H Br Na H NaNH2
- 1. BH3 @ THF
- 2. H2O2, OH-

SLIDE 17
A second strategy: a [(2C + 4C) + 2C] - 1C = 7C approach
H O H H H H Br H H H H H H Br H H + +

SLIDE 18 Forward synthesis:
H O H H H Br Na H H H H Br H H Na H H H H H H H O NaNH2 H2 Lindlar Pd HBr ROOR NaNH2 H2 Lindlar Pd
+

SLIDE 19
Using the general strategy of our second synthesis, can you think of a third way that makes heptanal without generating any other aldehydes as side-products?