Physics 2D Lecture Slides Lecture 27: Mar 8th Vivek Sharma UCSD - - PDF document
Physics 2D Lecture Slides Lecture 27: Mar 8th Vivek Sharma UCSD - - PDF document
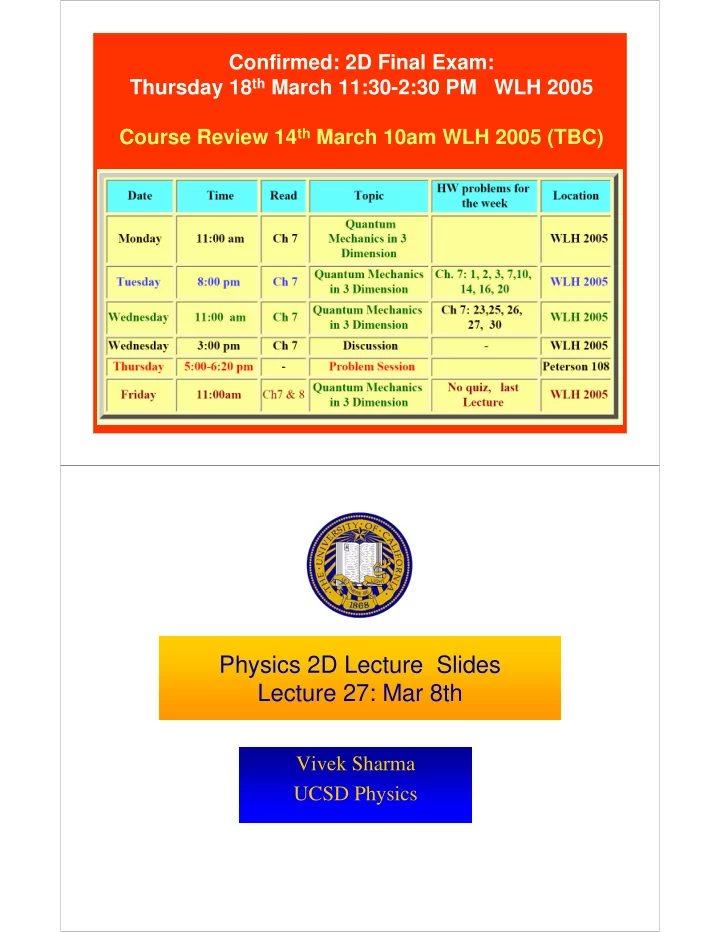
Confirmed: 2D Final Exam: Thursday 18 th March 11:30-2:30 PM WLH 2005 Course Review 14 th March 10am WLH 2005 (TBC) Physics 2D Lecture Slides Lecture 27: Mar 8th Vivek Sharma UCSD Physics Quiz 8 16 14 12 Frequency 10 8 6 4 2 0 0 1
Quiz 8
2 4 6 8 10 12 14 16
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
Grades Frequency
- Learn to extend S. Eq and its
solutions from “toy” examples in 1-Dimension (x) → three
- rthogonal dimensions
(r ≡x,y,z)
- Then transform the systems
– Particle in 1D rigid box 3D rigid box – 1D Harmonic Oscillator 3D Harmonic Oscillator
- Keep an eye on the number
- f different integers needed
to specify system 1 3 (corresponding to 3 available degrees of freedom x,y,z)
QM in 3 Dimensions
y z x
ˆ ˆ ˆ r ix jy kz = + +
Quantum Mechanics In 3D: Particle in 3D Box
Extension of a Particle In a Box with rigid walls 1D → 3D ⇒ Box with Rigid Walls (U=∞) in X,Y,Z dimensions
y y=0 y=L z=L z x
Ask same questions:
- Location of particle in 3d Box
- Momentum
- Kinetic Energy, Total Energy
- Expectation values in 3D
To find the Wavefunction and various expectation values, we must first set up the appropriate TDSE & TISE
U(r)=0 for (0<x,y,z,<L)
The Schrodinger Equation in 3 Dimensions: Cartesian Coordinates
2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2
Time Dependent Schrodinger Eqn: ( , , , ) ( , , , ) ( , , ) ( , ) .....In 3D 2 2 2 2 2 x y z t x y x y z t U x y z x t i m t m x m y m z m So ∂ ∂ ∂ ∇ ∂Ψ − ∇ Ψ + Ψ = ∂ − ∇ = + ⎛ ⎞ ⎛ ⎞ ∂ ∂ ∂ − − + − ⎜ ⎟ ⎜ ⎟ ∂ ∂ ∂ ⎝ ⎠ ⎝ +∂ ⎠ = + ∂ ∂
- x
2 x x
[K ] + [K ] + [K ] [ ] ( , ) [ ] ( , ) is still the Energy Conservation Eq Stationary states are those for which all proba [ ] = bilities so H x t E K x t z ⎛ ⎞= Ψ ⎟ ⎠ = ⎜ ⎝ Ψ
- i t
are and are given by the solution of the TDSE in seperable form: = (r)e This statement is simply an ext constant in time ( ension of what we , derive , , ) ( , ) d in case of x y z t r t
ω
ψ Ψ =Ψ
- 1D
time-independent potential
y z x
Particle in 3D Rigid Box : Separation of Orthogonal Spatial (x,y,z) Variables
1 2 3 1 2 2 3 2
in 3D: x,y,z independent of each ( , , ) ( ) ( ) ( ) and substitute in the master TISE, after dividing thruout by = ( ) ( ) (
- ( , , )
( ,
- ther , wr
, ) ( , , ) and ) ( , ite , ) n 2m x y z TISE x y z U x y z x y z E x y x y z x y z z ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ∇ = + =
- 2
2 1 2 1 2 2 2 2 2 2 3 2 3 2 1 2 2
( ) 1 2 ( ) This can only be true if each term is c
- ting that U(r)=0 fo
- nstant for all x,y,z
( ) 1 2 ( ) ( 2 r (0<x,y,z,<L) ( ) 1 2 ( ) z E Const m z z y m x m x x x m y y ψ ψ ψ ψ ψ ψ ψ ⎛ ⎞ ∂ − + ⎜ ⎟ ⎛ ⎞ ∂ + − = ⇒ ⎛ ⎞ ∂ − ⎜ ⎟ ∂ ⎝ ⎠ ∂ ⎝ ⎠ = ⇒ − ⎜ ⎟ ⎝ ⎠ ∂ ∂
- 2
2 3 3 3 2 2 2 2 2 2 1 1 2 2 1 2 3
) ( ) ; (Total Energy of 3D system) Each term looks like ( ) ( ) ; 2 With E particle in E E E=Constan 1D box (just a different dimension) ( ) ( ) 2 So wavefunctions t z E z m z y y E x E x y m ψ ψ ψ ψ ψ ∂ − = ∂ − ∂ = = = ∂ + + ∂
- 3
3 1 2 2 1
must be like , ( ) sin x , ( ) s ) s n in ( i y y k x k z k z ψ ψ ψ ∝ ∝ ∝
Particle in 3D Rigid Box : Separation of Orthogonal Variables
1 1 2 2 3 3 i
Wavefunctions are like , ( ) sin Continuity Conditions for and its fi ( ) sin y Leads to usual Quantization of Linear Momentum p= k .....in 3D rst spatial derivative ( ) s sin x ,
x i i
z k z n k x L y k p k ψ ψ π ψ π ψ ∝ ∝ ⇒ = ∝ =
- 1
2 3 2 2 1 3 1 2 2 2 2 2 2 3
; ; Note: by usual Uncertainty Principle argumen (n ,n ,n 1,2,3,.. ) t neither of n ,n ,n 0! ( ?) 1 Particle Energy E = K+U = K +0 = ) 2 ( m 2 (
z y x y z
n why p n L n mL p n L L p p p π π π ⎛ ⎞ ⎛ ⎞ = = ∞ ⎜ ⎟ ⎜ ⎛ ⎞ = ⎜ ⎟ ⎝ ⎠ ⎟ ⎝ ⎠ ⎝ = ⎠ + + =
- 2
2 2 1 2 3 2 1 2 3 2 1 E i 3
- 3
1
) Energy is again quantized and brought to you by integers (independent) and (r)=A sin (A = Overall Normalization Co sin y (r) nstant) (r,t)= e [ si n ,n ,n sin x sin x ys n in ]
t
k n n k A k k k k z z ψ ψ + + = Ψ
- E
- i
e
t
Particle in 3D Box :Wave function Normalization Condition
3 * 1 1 2 1 x,y, E E
- i
- i
2 E E i i * 2 2 2 2 2 3 * 3 z 2
(r) e [ sin y e (r) e [ s (r,t)= sin ] (r,t)= sin ] (r,t) sin x sin x sin x in y e [ si Normalization Co (r,t)= sin ] ndition : 1 = P(r)dx n y dyd 1 z
t t t t
k z k k k A k A k A k z k k A z ψ ψ Ψ Ψ Ψ ⇒ Ψ = = =
∫∫∫
- L
L L 2 3 3 E 2 2 2 1 2 3 x=0 y= 2 2
- 1
z 3 i 2 =0
sin x dx s sin y dy sin z dz = ( 2 2 2 2 2 an r,t)= d [ s sin i i e x y n ] n
t
L k L L A A k L k k k k z L ⎛ ⎞⎛ ⎞⎛ ⎞ ⎜ ⎟⎜ ⎟⎜ ⎟ ⎜ ⎟⎜ ⎟⎜ ⎟ ⎝ ⎠⎝ ⎠⎝ ⎠ ⎡ ⎤ ⎡ ⎤ ⇒ = ⎢ ⎥ ⎢ ⎥ ⎣ ⎦ ⎣ ⎦ Ψ
∫ ∫ ∫
- Particle in 3D Box : Energy Spectrum & Degeneracy
1 2 3
2 2 2 2 2 n ,n ,n 1 2 3 i 2 2 111 2 2 2 211 121 112 2 2
3 Ground State Energy E 2 6 Next level 3 Ex E ( ); n 1, 2,3... , 2 s cited states E = E E 2 configurations of (r)= (x,y,z) have Different ame energy d
i
mL mL n n n n mL π π ψ ψ π = + + = ∞ ≠ = ⇒ = = ⇒
- egeneracy
y y=L z=L z x x=L
2 2 211 121 112 2
Degenerate States 6 E = E E 2mL π = =
- x
y z E211 E121 E112 ψ E111 x y z ψ Ground State
Probability Density Functions for Particle in 3D Box
Same Energy Degenerate States Cant tell by measuring energy if particle is in 211, 121, 112 quantum State
Source of Degeneracy: How to “Lift” Degeneracy
- Degeneracy came from the
threefold symmetry of a CUBICAL Box (Lx= Ly= Lz=L)
- To Lift (remove) degeneracy
change each dimension such that CUBICAL box Rectangular Box
- (Lx≠ Ly ≠ Lz)
- Then
2 2 2 2 2 2 3 1 2 2 2 2
2 2 2
x y z
n n n E mL mL mL π π π ⎛ ⎞ ⎛ ⎞ ⎛ ⎞ = + + ⎜ ⎟ ⎜ ⎟ ⎜ ⎟ ⎜ ⎟ ⎝ ⎠ ⎝ ⎠ ⎝ ⎠
Energy The Hydrogen Atom In Its Full Quantum Mechanical Glory
2
( ) kZe U r r =
2 2 2
By example of particle in 3D box, need to use seperation of variables(x,y,z) to derive 3 in 1 1 This approach will dependent d ( ) M iffer
- re compli
- ential. eq
cated form of U than bo get x ns. U r r x y z ∝ = ⇒ + +
2 2 2 2
To simplify the situation, use appropriate variables Independent Cartesian (x,y,z)
- Inde. Spherical Polar (r, ,
very ugly since we have a "conjoined triplet" Instead of writing Laplacian ) x y θ φ ∂ ∂ ∇ = + ∂ ∂ →
2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2
, write 1 sin TISE for (x,y,z)= (r, , ) become 1 r 1 (r, , ) (r, , ) r s 1 2m + 1 = s (E-U(r)) in si si n 1 sin s (r, , ) n in r r r r z r r r r r r θ θ θ θ θ θ θ θ φ ψ ψ θ ψ θ φ ψ θ φ ψ θ φ θ φ θ φ ∂ ∂ ⎛ ⎞ ⎜ ⎟ ∂ ∂ ⎝ ⎠ ∂ ∂ ⎛ ∂ ∂ ⎛ ⎞ ⎜ ⎟ ∂ ∂ ∂ + ∂ ∂ ∂ ∂ ∂ ⎞ ∇ + + ⎜ ⎟ ∂ ∂ ⎝ ⎠ ∂ ∂ ⎛ ⎞ + + ⎜ ∂ ⎠ ⎝ ⎟ ∂ ⎝ ⎠
- !!!! fun!!!
(r, , ) =0 ψ θ φ
r
Spherical Polar Coordinate System
2
( sin ) Vol ( )( ) = r si ume Element dV n dV r d rd dr drd d θ φ θ θ θ φ =
Don’t Panic: Its simpler than you think !
2 2 2 2 2 2 2 2 2 2 2 2
1 2m + (E-U(r)) sin Try to free up las 1 (r, , ) =0 r all except This requires multi t term fro plying thruout by sin si 1 sin si si si m n n n r r r r r r r r r ψ ψ ψ ψ θ φ φ θ ψ θ θ θ θ θ θ θ θ φ ∂ ∂ ⎛ ⎞ ⎜ ⎟ ∂ ∂ ⎝ ⎠ ⇒ ∂ ∂ ⎛ ⎞ ⎜ ⎟ ∂ ∂ ⎝ ⎠ ∂ ∂ ⎛ ⎞ + + ⎜ ⎟ ∂ ∂ ⎝ ⎠ ∂ + ∂ ∂ ∂
- 2
2 2 2 2 2
2m ke + (E+ ) r (r, , )=R(r). ( ). ( ) Plug it into the TISE above & divide thruout by (r, , )=R(r). ( ). sin =0 For Seperation of Variables, W rite ( , , ) r Note tha ( t : ) n r r φ ψ θ φ θ φ ψ ψ θ ψ ψ θ φ φ θ θ θ φ θ ∂ ⎛ ⎞ + ⎜ ⎟ ∂ ⎝ ⎠ ∂ ∂ ∂ Θ Ψ Θ Φ Φ ∂
- 2
2 2 2 2 2 2 2
( ). ( ) ( , , ) ( ) ( ) ( , , ) ( ) ( ) s R(r) r ( ) when substituted in TI in sin =0 Rearrange by ta sin king the sin SE ( ) 1 2m ke + (E+ ) r r R r r R r R r r R r r θ φ θ φ φ θ θ φ θ θ θ θ φ φ θ θ φ θ θ θ θ = Θ Φ ∂Ψ = Φ ∂ ∂Ψ = Θ ∂ ∂ ∂ ⎛ ∂ ∂Θ ⎛ ⎞ + + ⎜ ⎟ Θ ⎞ ⎜ ∂ ∂ ⎝ ⎟ ∂ ∂ ∂ ∂ ∂Θ ⇒ ∂ ∂Φ ∂ ∂ ∂ ⎝ Φ ⎠ ⎠ Φ
- 2
2 2 2 2 2 2 2 2
2m ke 1 + (E+ ) r LHS is fn. of r, & RHS is fn of
- nly , for equality to be true for all r,
, LHS= constant = RH term on RHS sin s S = sin sin m in =-
l
R r r R r r φ θ φ θ φ θ θ φ θ θ θ θ ∂ ∂ ⎛ ⎞ ⎜ ⎟ ∂ Φ Φ ∂ ⇒ ∂ ∂Θ ⎛ ⎞ + ⎜ ⎟ Θ ∂ ∂ ⎝ ⎝ ⎠ ⎠ ∂ ∂
2 2 2 2 2 2 2 2 2
sin sin =m Divide Thruout by sin and arrange all terms with r aw Now go break up LHS to seperate the terms... r .. 2m ke LHS: + (E+ ) a & sin si y from r 1 n
l
R r r r r r R r R θ θ θ θ θ θ θ θ θ ∂ ∂Θ ⎛ ⎞ + ⎜ ⎟ Θ ∂ ∂ ⎝ ⎠ ∂ ∂ ⎛ ∂ ⎞ ∂ ∂ ⎜ ⎟ ∂ ∂ ⎝ ⎠ ⇒
- 2
2 2 2 2
m 1 sin sin sin Same argument : LHS is fn of r, RHS is fn of , for them to be equal for a LHS = const = 2m ke (E+ )= r What do we have after shuffl ll r, = ( in 1) g RHS
l
l l R r r θ θ θ θ θ θ θ ∂ ∂Θ ⎛ ⎞ − ⎜ ⎟ ⎛ ⎞ + Θ ∂ ∂ ⎝ ⎠ ⇒ + ⎜ ⎟ ∂ ⎝ ⎠
- 2
2 2 2 2 2 2 2 2 2 2
! m 1 sin ( 1) ( ) 0.....(2) sin sin ............... d ..(1) 1 2m ke ( 1) (E+ )- ( ) 0....( m 0. 3 . ) r T
l l
d R r l l r R r r dr d d l d d r l d r θ φ θ θ θ θ θ ⎡ ⎤ Θ ⎛ ⎞ + + − Θ ⎡ ⎤ ∂ + ⎛ ⎞ + = ⎜ ⎟ ⎢ ⎥ ∂ ⎝ ⎠ ⎣ ⎦ = ⎜ ⎟ ⎢ ⎥ ⎝ ⎠ ⎣ ⎦ Φ + Φ =
- hese 3 "simple" diff. eqn describe the physics of the Hydrogen atom.
All we need to do now is guess the solutions of the diff. equations Each of them, clearly, has a different functional form
Solutions of The S. Eq for Hydrogen Atom
2 2 2
d The Azimuthal Diff. Equation : m Solution : ( ) = A e but need to check "Good Wavefunction Condition" Wave Function must be Single Valued for all ( )= ( 2 ) ( ) = A e
l l
l im im
d
φ φ
φ φ φ φ φ π φ Φ + Φ = Φ ⇒ Φ Φ + ⇒ Φ
( 2 ) 2 2
A e 0, 1, 2, 3....( ) The Polar Diff. Eq: Solutions : go by the name of "Associated Legendre Functions" 1 m sin ( 1) ( ) sin si Quantum # n
l
l im l
d d Magnetic l d m l d
φ π
θ θ θ θ θ θ
+
⎡ ⎤ Θ ⎛ ⎞ + + − Θ = ⎜ ⎟ ⎢ ⎥ ⎝ ⎠ = ⎣ ⎦ ⇒ = ± ± ±
- nly exist when the integers and
are related as follows 0, 1, 2, 3.... : Orbital Q ; ua p nt
- sit
um N ive numb umber er 1 For 0, =0 ( ) = ; 2 For
l l l
l m l l l m l m θ = ± ± ± ± = = ⇒ Θ
2
1, =0, 1 Three Possibilities for the Orbital part of wavefunction 6 3 [ 1, 0] ( ) = cos [ 1, 1] ( ) = sin 2 2 10 [ 2, 0] ( ) = (3cos 1).... so on and so forth (see book) 4
l l l l
l m l m l m l m and θ θ θ θ θ θ = ± ⇒ = = ⇒ Θ = = ± ⇒ Θ = = ⇒ Θ −
Φ
Solutions of The S. Eq for Hydrogen Atom
2 2 2 2 2 2 2 2
1 2m ke ( 1) The Radial Diff. Eqn: (E+ )- ( ) r : Associated Laguerre Functions R(r), Solutions exist only if:
- 1. E>0 or has negtive values given by
ke 1 E=- 2a d R r l l r R r r dr r r Solutions n ⎡ ⎤ ∂ + ⎛ ⎞ + = ⎜ ⎟ ⎢ ⎥ ∂ ⎝ ⎠ ⎦ ⎝ ⎣ ⎛
- 2
2
- 2. And when n = integer such that
0,1,2,3,4,,,( 1) n = principal Quantum # or the "big daddy" qunatu To ; Bohr Summa m # : The hy Rad r droge ize n ius atom a l mke n = − ⎞ = = ⎜ ⎟ ⎠
- n = 1,2,3,4,5,....
0,1,2,3,,4....( 1) m 0, 1, 2, 3,. Quantum # appear only in Trappe is brought to you d systems The Spati by the letters al Wave Function o f the Hydrogen Atom ..
l
l n l ∞ = − = ± ± ± ±
l
m
( , , ) ( ) . ( ) . ( ) Y (Spherical Harmonics)
l l
m nl lm nl l
r R r R θ φ θ φ Ψ = Θ Φ =
Radial Wave Functions & Radial Prob Distributions
- r/a
3/2 r
- 2
a 3/2 2 3 2 3/2
R(r)= 2 e a 1 r (2 n 1 0 0 2 0 0 3 0 0
- )e
a 2 2a 2 r (27 18 2 ) a 81 3a
l r a
r e a l m
−
− +
n=1 K shell n=2 L Shell n=3 M shell n=4 N Shell
…… l=0 s(harp) sub shell l=1 p(rincipal) sub shell l=2 d(iffuse) sub shell l=3 f(undamental) ss l=4 g sub shell ……..
Symbolic Notation of Atomic States in Hydrogen
2 2 4 4 2 ( 0) ( 1) ( 2) ( 3) ( 4 3 3 3 ) ..... 1 4 3 1 s p s l p l d l f l g l s l s d n p s p = = = → = = ↓ 5 5 5 5 4 5 4 5 s p d f g d f
Note that:
- n =1 non-degenerate system
- n1>1 are all degenerate in l and ml.
All states have same energy But different spatial configuration
2 2
ke 1 E=- 2a n ⎛ ⎞ ⎜ ⎟ ⎝ ⎠
Facts About Ground State of H Atom
- r/a
3/2
- r/a
100
2 1 1 ( ) e ; ( ) ; ( ) a 2 2 1 ( , , ) e ......look at it caref
- 1. Spherically s
1, 0, ymmetric no , dependence (structure)
- 2. Probab
ully i
l
n l r r a m R θ φ π θ φ π θ φ ⇒ = Θ = Φ = Ψ = ⇒ = = =
2 2 100 3
Likelihood of finding the electron is same at all , and depends only on the radial seperation (r) between elect 1 lity Per Unit Volume : ( , ron & the nucleus. 3 Energy ,
- f Ground ta
) S
r a
r e a θ π θ φ φ
−
Ψ =
2
ke te =- 13.6 2a Overall The Ground state wavefunction of the hydrogen atom is quite Not much chemistry or Biology could develop if there was
- nly the ground state of the Hydrogen Ato
We ne m e ! boring eV = − d structure, we need variety, we need some curves!
Interpreting Orbital Quantum Number (l)
2 RADIAL ORBITAL RADIAL ORBI 2 2 2 2 2 2 2 2 TAL 2 2
K 1 2m ke ( 1) Radial part of S.Eq K ;substitute this form for n: (E+ )- ( ) r For H Atom: E = E K K K + U = 1 2m ( 1)
- 2m
d dR r l l r R r r dr dr r d dR l l r r dr dr r ke r ⎡ ⎤ + ⎛ ⎞+ = ⎜ ⎟ ⎢ ⎥ ⎝ ⎠ ⎣ ⎦ + ⎛ ⎞+ ⎜ + ⎟ − + ⎝ ⎠
- [
]
ORBITAL RADIAL OR 2 2 2 2 B 2 2
( ) ( 1) Examine the equation, if we set get a diff. eq. in r 2m 1 2m ( ) 0 which Further, we also kno depends only on radi K K us w r of orb K tha i t t R r l l then r d dR r R r r dr dr ⎡ ⎤ = ⎢ ⎥ ⎣ ⎦ + ⎛ ⎞+ = ⎜ ⎟ ⎝ ⎠ =
- 2
2 ITAL ORBIT
- rb
2 AL 2 2 ORBITAL 2 2
L= r p ; |L| =mv r ( 1 1 ; K 2 2 Putting it all togather: K magnitude of ) | | ( 1) 2m Since integer=0,1,2,3...( A n-1) angular mome ng. 2 nt Mom
- rbit
l l L l l r l L mv mr L posi mr tive × ⇒ + = = + = = = ⇒ = ⇒
- um|
| ( 1) | | ( 1) : QUANTIZATION OF Electron's Angular Momentum L l l discrete values L l l = + = = +
- p
r L Magnetic Quantum Number ml
(Right Hand Rule) QM: Can/Does L have a definite direction Classically, direction & Magnitud ? Proof by Negat ˆ Suppose L was precisely known/defined (L || z) e of L S always well defi n ed : n io L r p = ×
- 2
z z z
Electron MUST be in x-y orbit plane z = 0 ; , in Hydrogen atom, L can not have precise measurable ince Uncertainty Principle & An p p ; !!! gular Momentum value : L 2 p z E L r p So m φ = × ⇒ ⇒ ∆ ∆ ∆ ⇒ ∆ ∞ ∆ ∞ ∆ =
- ∼
∼ ∼
- ∼
Z Z
The Z compo Arbitararily nent of L vector spins around Z axis (precesses). |L | ; 1, 2, 3... ( 1) It picking Z axis as a referen L :| L | ca | | (always) s n in ce directi c
- :
e n
l l l
m Note L m l m l l < = = ± ± ± ± < +
- Z
never be that |L | ( 1) (breaks Uncertainty Principle) So you see, the dance has begun !
l
m l l = = +