SLIDE 1
Naming double bond-containing substituents IUPAC: alkenyl - - PowerPoint PPT Presentation
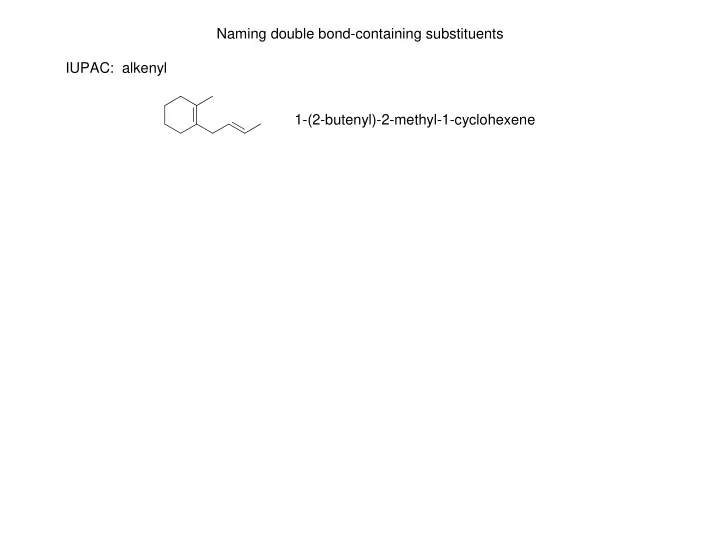
Naming double bond-containing substituents IUPAC: alkenyl - - PowerPoint PPT Presentation
Naming double bond-containing substituents IUPAC: alkenyl 1-(2-butenyl)-2-methyl-1-cyclohexene Naming double bond-containing substituents IUPAC: alkenyl 1-(2-butenyl)-2-methyl-1-cyclohexene Some common double bond-containing substituents:
SLIDE 2
SLIDE 3
Naming double bond-containing substituents IUPAC: alkenyl 1-(2-butenyl)-2-methyl-1-cyclohexene Some common double bond-containing substituents: vinyl (ethenyl) 1-methyl-4-vinyl-1-cyclohexene allyl (2-propenyl) 1-allyl-4-vinyl-1-cyclohexene
SLIDE 4
Naming double bond-containing substituents IUPAC: alkenyl 1-(2-butenyl)-2-methyl-1-cyclohexene Some common double bond-containing substituents: vinyl (ethenyl) 1-methyl-4-vinyl-1-cyclohexene allyl (2-propenyl) 1-allyl-4-vinyl-1-cyclohexene phenyl 4,4-dimethyl-1-phenyl-1-cycloheptene
SLIDE 5
Naming double bond-containing substituents IUPAC: alkenyl 1-(2-butenyl)-2-methyl-1-cyclohexene Some common double bond-containing substituents: vinyl (ethenyl) 1-methyl-4-vinyl-1-cyclohexene allyl (2-propenyl) 1-allyl-4-vinyl-1-cyclohexene phenyl 4,4-dimethyl-1-phenyl-1-cycloheptene benzyl (phenylmethyl) (R)-1-benzyl-4-methyl-4-phenyl-1-cycloheptene
SLIDE 6
Cl
When the parent chain contains multiple C=C, use a multiplicative prefix before “ene”: (S)-6-chloro-4-methyl-2,4-heptadiene
SLIDE 7
H H H H H H H H
Alenes are classified according to their degree of substitution of the C=C functional group monosubstituted disubstituted terminally disubstituted trisubstituted tetrasubstituted
SLIDE 8
C H3 CH3 C H2 CH2 CH3 + . CH3 . :CH2 + :CH2
Csp2
- Csp2
- H1s
H1s H1s H1s
How strong is a C=C ? Is it twice the strength of a C–C? HDBE, kcal/mol 83 1 σ 146 1 σ + 1 π
- HBDE (1 σ + 1 π) - HBDE (1 σ ) = HBDE (1 π)
146
- 83 = 63
SLIDE 9
cis-alkenes and trans-alkenes Consider all the isomers of the alkene C4H8: are these conformational isomers?
- No! They are isomers that differ by the relative positions of the substituent methyl
groups in space.
- They cannot be converted into each other without breaking a bond.
- Therefore, they are diastereoisomers.
- Why does rotation about the C=C break a bond?
SLIDE 10
C C C H3 CH3 H H C CH3 H C C H3 H C C H CH3 H CH3
π bond no π bond π bond reforms These diastereoisomers are named just like disubstituted cycloalkanes:
- Identical groups on the same side of the C=C (that is, with a 0 dihedral angle)
are cis-alkenes
- Identical groups on opposite sides of the C=C (that is, with a 180 dihedral
angle) are trans-alkenes
SLIDE 11
O OH H H Cl OH CH3 Br
trans-2-butene cis-2-butene trans-cinnamic acid ?
SLIDE 12
The E,Z system of naming diastereomeric alkenes:
- Use the Cahn-Ingold-Prelog “Priority Rules”
- 1. For each C of the C=C, rank the priorities of the 2 atoms attached, according to
atomic number.
- 2. If the 2 atoms attached are identical, rank the atoms attached to those 2 atoms.
Priority of the entire substituent is determined by the first point of difference.
- 3. If identical priority numbers are on the same side of the C=C, the alkene is a
(Z)-alkene.
- 4. If identical priority numbers are on opposite sides of the C=C the alkene is a
(E)-alkene.
SLIDE 13
C X Y Z C X X C Y Z
For comprison purposes, identical atoms must be compared in the same hybridization state:
- The “Phantom atom” Rules
- 1. For naming purposes, a π bonded atom is considered to replicated by a phantom
σ bond to a phantom atom.
- 2. Rule 1 is applied to both atoms of the π bond.
- 3. Rules 1 and 2 apply to each π bond of multiply bonded atoms.
- 4. All other things being equal, phantom atoms rank below real atoms of the same
atomic number.
SLIDE 14
Cl Cl C4 C3 C C C C Cl Cl H H H H H H H 1 2 C C 2 1
An example: What is the IUPAC name of ?
- 1. The longest chain containing both C=C bonds is 5:
pentadiene
- 2. Number the chain to give the C=C bonds the lowest locants:
1,3-pentadiene
- 3. Add substituents:
1,5-dichloro-3-ethyl-4-methyl-1,3-pentadiene
- 4. Add stereodesignator for the C1=C2 bond:
(1E)-1,5-dichloro-3-ethyl-4-methyl-1,3-pentadiene
- 5. Add stereodesignator for the C3=C4 bond:
(1E,3E)-1,5-dichloro-3-ethyl-4-methyl-1,3-pentadiene
SLIDE 15
Another example using the phantom atom rule: Is this an E or a Z alkene?
C C O C O H H H H H
SLIDE 16
Another example using the phantom atom rule: Is this an E or a Z alkene? It’s an E alkene.
1 2 1 2
SLIDE 17
Thermodynamic stabilities of isomeric alkenes: Consider C4H8 + 6 O2 4 CO2 + 4 H2O H°rxn = H°rxn kcal/mol CO2 + H2O
SLIDE 18
The substitution - elimination continuum
what factors influence the balance between substitution and elimination for a given class of haloakanes?
SLIDE 19
Primary haloalkanes:
- SN2 for all Z-
- Increased steric hindrance in ‡ increases the proportion of E2
P
- branching in haloalkane
P
strong, bulky Lewis bases
SLIDE 20
Secondary haloalkanes:
- SN2 for all Z- less basic than HO-
- E2 for all Z- more basic than HO-
- Increased steric hindrance in ‡ increases the proportion of E2
P
- branching in haloalkane
P
strong, bulky Lewis bases
- SN1 or E1 when Z: is neutral and the solvent
CA of Nu: pKa H-CN 9.2 H-OH 15.7 H-CH2CH3 50
SLIDE 21
Tertiary haloalkanes:
- E2 for all Z-
- SN1 or E1 for most Z: and the anions of strong CA
Generally, regardless of the identity of Z: or Z- :
- SN is favored at lower temperatures
- E is favored at higher temperatures