LIGAND EFFECT: CHELATING prim- AMINE OH OH [RuCl 2 (hmb)] 2 amine - - PowerPoint PPT Presentation
LIGAND EFFECT: CHELATING prim- AMINE OH OH [RuCl 2 (hmb)] 2 amine - - PowerPoint PPT Presentation
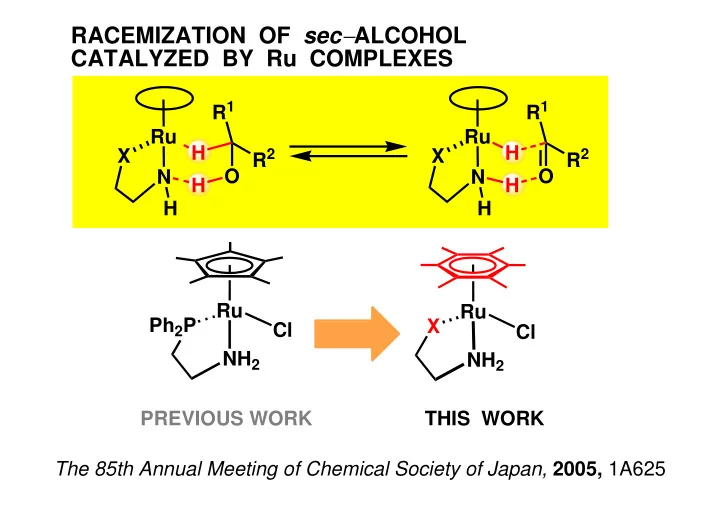
RACEMIZATION OF sec ALCOHOL CATALYZED BY Ru COMPLEXES R 1 R 1 Ru Ru H H X X R 2 R 2 N O N O H H H H Ru Ru Ph 2 P X Cl Cl NH 2 NH 2 PREVIOUS WORK THIS WORK The 85th Annual Meeting of Chemical Society of Japan, 2005,
LIGAND EFFECT: CHELATING prim-AMINE
KOt-C4H9 toluene, 30 °C OH OH [RuCl2(hmb)]2 amine ligand HS NH2 HO NH2 TsN NH2 H 7 14 76 HO NH2 HO S NH2 O O O 58 86 SN chelate ee, %
- ther
>99% ee PhS NH2 91 HS NMe2 99 24 h S/C = 100 Cl Ru Ru Cl Cl Cl [RuCl2(hmb)]2 ee, % amine ligand:
LIGAND EFFECT: ARENE
KOt-C4H9 toluene, 30 °C OH OH [RuCl2(arene)]2 99 99 7 >99% ee 24 h S/C = 100 92 97 arene ligand ee, % arene ligand ee, % Cl Ru Ru Cl Cl Cl [RuCl2(arene)]2 Rn Rn HS(CH2)2NH2 94
REACTION OF [RuCl2(hmb)]2 WITH SN LIGAND
1/2 [RuCl2(hmb)]2 + HS NH2 R1 = 0.057 wR2 = 0.148 1.98 (s, 18H) 2.17 (s, 18H)
1H NMR (δ, CDCl3) :
P-1 (#2) + 1/2 KOt-C4H9 2-propanol 30 °C 91% yield Ru(1) –N(1) –C(14) 110.2 Bond angles (°) Ru(1) –S(2) –Ru(2) 99.7 Ru(1) –S(1) –Ru(2) 100.6 S Ru Ru S H2N NH3 Cl Cl2 – Isolation of catalyst precursor – 1/2 Ru(1) –Ru(2) 3.6790 Bond lengths (Å)
EFFECT OF STOICHIOMETRY AND REACTION CONDITIONS
[RuCl2(hmb)]2 Ru S NH2 S HS NH2 NH2 P-1 (#2) R1 = 0.120 wR2 = 0.318 Bond lengths (Å) Ru(1) –S(1) 2.383 Ru(1) –S(2) 2.409 Bond angles (°) Ru(1) –N(1) –C(14) 115.1 Ru(1) –S(2) –C(15) 113.4 Ru(1) –S(1) –C(13) 99.7
1H NMR (δ, CDCl3): 2.01(s, 18H)
Ru (excess) SN (excess) 2-propanol KOH THF CH3ONa R1 = 0.079 wR2 = 0.188 P-1 (#2) 2.27 (s, 36H)
1H NMR (δ, CDCl3):
Bond lengths (Å) Bond angles (°) Ru(1) –Ru(2) 2.7328 Ru(1) –S(1) –Ru(2) 70.51 Ru(1) –N(1) –Ru(2) 82.5 –12.3 (s, 1H) 78% yield 68% yield 30 °C 30 °C Ru Ru N H S H Cl