Ion Neutralization at Metal Surface
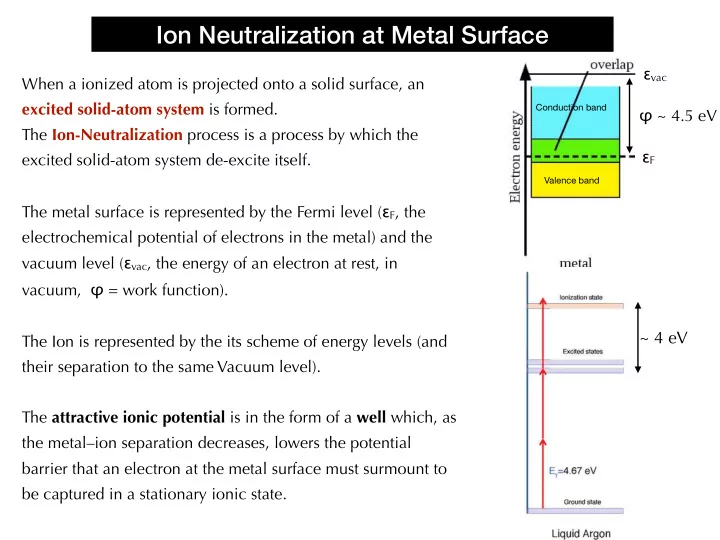
When a ionized atom is projected onto a solid surface, an excited solid-atom system is formed. The Ion-Neutralization process is a process by which the excited solid-atom system de-excite itself. The metal surface is represented by the Fermi level (εF, the electrochemical potential of electrons in the metal) and the vacuum level (εvac, the energy of an electron at rest, in vacuum, φ = work function). The Ion is represented by the its scheme of energy levels (and their separation to the same Vacuum level). The attractive ionic potential is in the form of a well which, as the metal–ion separation decreases, lowers the potential barrier that an electron at the metal surface must surmount to be captured in a stationary ionic state.
Valence band Conduction band