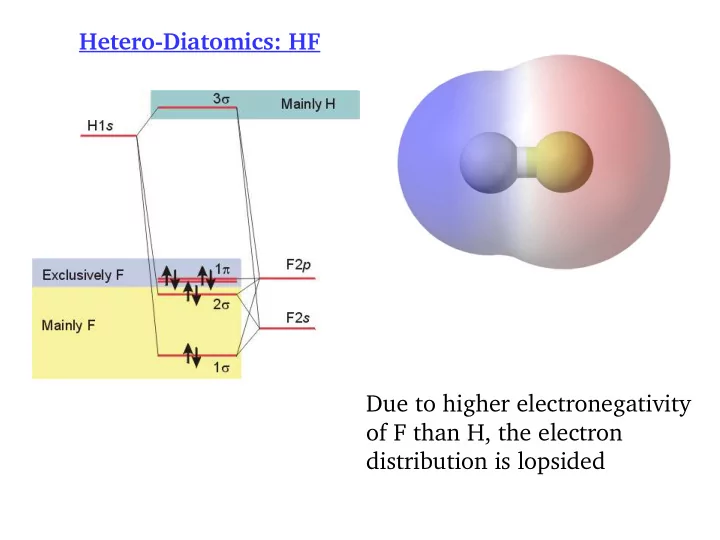
SLIDE 1 Hetero-Diatomics: HF Due to higher electronegativity
distribution is lopsided

SLIDE 2
Hetero-Diatomics: HCl For Cl 3p states close in energy to the 1s of H

SLIDE 3
Hetero-Diatomics: HBr For Cl 4p states close (higher) in energy to the 1s of H

SLIDE 4
Hetero-Diatomics: CO

SLIDE 5
Hybridization Linear combination of atomic orbitals within an atom leading to more effective bonding 2s 2pz 2px 2py 2px 2py α 2s-β 2pz α 2s+β 2pz The coefficients α and β depend on field strength
Hybridization is close to VBT approach. Use of experimental information All hybridized orbitals are equivalent and are ortho-normal to each other

SLIDE 6 Contribution from s=0.5; contribution from p=0.5 Have to normalize each hybridized orbital 2 equivalent hybrid orbitals
shape (directions different)
Linear geometry with Hybridized atom at the center s and p orbital of the Same atom! Not same as S (overlap)
s+p (sp)Hybridization = −
1
1 2
s p
ψ ψ ψ = +
2
1 2
s p
ψ ψ ψ

SLIDE 7
s+p (sp)Hybridization

SLIDE 8
The other p-orbitals are available for π bonding s+p (sp)Hybridization

SLIDE 9
Molecular orbitals of BeH2

SLIDE 10 Molecular orbitals of BeH2
( ) ( ) ( ) ( )
* 4 2 3 1 1 * 2 2 1 1 1 3 2 4 1 1 1 2 2 1 1
A B A B A B A B
H H u Be s s H H g Be s s H H u Be s s H H g Be s s
pz s pz s
c c c c c c c c
σ σ σ σ
ϕ ψ ψ ψ ϕ ψ ψ ψ ϕ ψ ψ ψ ϕ ψ ψ ψ = − − = − + = + − = + +

SLIDE 11 Molecular orbitals & Hybridization in BeH2
g u σ σ
ϕ ϕ −
g u σ σ
ϕ ϕ +
( )
3 2 4 1 1
A B
H H u Be s s
pz
c c
σ
ϕ ψ ψ ψ = + −
( )
1 2 2 1 1
A B
H H g Be s s
s
c c
σ
ϕ ψ ψ ψ = + +

SLIDE 12
s+2p (sp2)Hybridization px and py can be combined with s to get three 3 equivalent hybrids at 120o to each other x y
30o 30o
x y
30o 30o
x y
30o 30o
+
x
s p − + cos(60) cos(30)
x y
s p p − − cos(60) cos(30)
x y
s p p

SLIDE 13 The other p-orbital are available for π bonding s+2p (sp2)Hybridization
1 2 3
1 2 3 3 1 1 1 3 2 6 1 1 1 3 2 6
s px py s px py s px py
ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ = + × + = + − = − −

SLIDE 14
s+2p (sp2)Hybridization

SLIDE 15 How to calculate the coefficients? Use orthogonality of hybrid orbitals and normalization conditions There is no unique solution
s+3p (sp3)Hybridization
1 2 3 4
1 1 1 1 2 2 2 2 1 1 1 1 2 2 2 2 1 1 1 1 2 2 2 2 1 1 1 1 2 2 2 2
s px py pz s px py pz s px py pz s px py pz
ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ = + + + = − − + = + − − = − + −
1 2 3 4
1 3 2 2 1 2 1 2 3 2 3 1 1 1 1 2 6 2 2 3 1 1 1 1 2 6 2 2 3
s px py pz s px py pz s px py pz s px py pz
ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ ψ = + × + × + = + + × − = − + − = − − −

SLIDE 16
s+3p (sp3)Hybridization No other p-orbital is available for π bonding

SLIDE 17 Asymmetrical Hybridization: Water Molecule Electronic Configuration of ‘O’ atom: 1s22s22pz22px12py1 104.5o φ φ = + = +
1 1 2 2 3 4
1 2 1 2
A B
H xO H yO
c s c p c s c p ψ ψ = + + = + +
1 1 2 3 2 4 5 6
( ) 2 2 2 ( ) 2 2 2
x y x y
O a s a p a p O a s a p a p ψ ψ = × + × + × = × − × + ×
1 2
( ) 0.45 2 0.71 2 0.55 2 ( ) 0.45 2 0.71 2 0.55 2
x y x y
O s p p O s p p

SLIDE 18
Asymmetrical Hybridization: Water Molecule Electronic Configuration of ‘O’ atom: 1s22s22pz22px12py1 Form bonds with two Hydrogen atoms Two lone pair electrons ψ ψ ψ ψ = × + × + × = × − × + × = × − × =
1 2 3 4
( ) 0.45 2 0.71 2 0.55 2 ( ) 0.45 2 0.71 2 0.55 2 ( ) 0.77 2 0.63 2 ( ) 2
x y x y y z
O s p p O s p p O s p O p

SLIDE 19
Asymmetrical Hybridization: Water Molecule Electronic Configuration of ‘O’ atom: 1s22s22pz22px12py1 Form bonds with two Hydrogen atoms Two lone pair electrons NOT Equivalent! ψ ψ ψ ψ = × + × + × = × − × + × = × − × =
1 2 3 4
( ) 0.45 2 0.71 2 0.55 2 ( ) 0.45 2 0.71 2 0.55 2 ( ) 0.77 2 0.63 2 ( ) 2
x y x y y z
O s p p O s p p O s p O p

SLIDE 20
Asymmetrical Hybridization: Water Molecule Electronic Configuration of ‘O’ atom: 1s22s22pz22px12py1 Form bonds with two Hydrogen atoms Two lone pair electrons NOT Equivalent! 104.5o ψ ψ = × − × + × = × − × − ×
3 4
( ) 0.55 2 0.45 2 0.71 2 ( ) 0.55 2 0.45 2 0.71 2
y z y z
O s p p O s p p ψ ψ ψ ψ = × + × + × = × − × + × = × − × =
1 2 3 4
( ) 0.45 2 0.71 2 0.55 2 ( ) 0.45 2 0.71 2 0.55 2 ( ) 0.77 2 0.63 2 ( ) 2
x y x y y z
O s p p O s p p O s p O p

SLIDE 21
Asymmetrical Hybridization: Water Molecule Form bonds with two Hydrogen atoms Lone pair electrons
Lone pair electrons have more ‘s’ character than the bonding orbitals
ψ ψ ψ ψ = × + × + × = × − × + × = × − × + × = × − × − ×
1 2 3 4
( ) 0.45 2 0.71 2 0.55 2 ( ) 0.45 2 0.71 2 0.55 2 ( ) 0.55 2 0.45 2 0.71 2 ( ) 0.55 2 0.45 2 0.71 2
x y x y y z y z
O s p p O s p p O s p p O s p p

SLIDE 22 Asymmetrical Hybridization: Water Molecule Bond pairs Lone pairs ψ ψ ψ ψ = × + × + × = × − × + × = × − × + × = × − × − ×
1 2 3 4
( ) 0.45 2 0.71 2 0.55 2 ( ) 0.45 2 0.71 2 0.55 2 ( ) 0.55 2 0.45 2 0.71 2 ( ) 0.55 2 0.45 2 0.71 2
x y x y y z y z
O s p p O s p p O s p p O s p p
0.0 0.5 1.0 θ =104.47
0.71 0.55 0.71 0.55
θ'=tan
=37.76

SLIDE 23
s-p3-d2 & s-p3-d Hybridization Sp3d2 Octahedral Sp3d Trignoal bipyramidal

SLIDE 24
Hybridization and Geometry

SLIDE 25
Do Orbitals Really Exist? Tomographic image of HOMO of N2
Nature; Volume 342; Year 2004; 867-871