SLIDE 1
Growth in Known Compounds 70,000,000 63,175,733 60,000,000 - - PowerPoint PPT Presentation
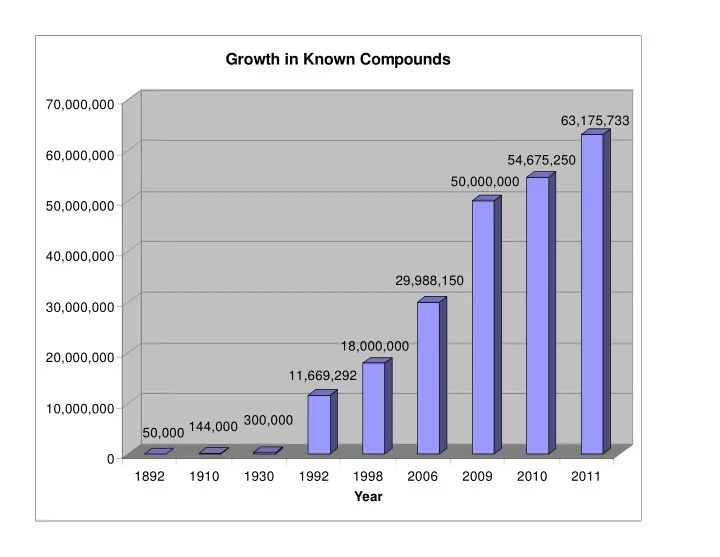
Growth in Known Compounds 70,000,000 63,175,733 60,000,000 - - PowerPoint PPT Presentation
Growth in Known Compounds 70,000,000 63,175,733 60,000,000 54,675,250 50,000,000 50,000,000 40,000,000 29,988,150 30,000,000 18,000,000 20,000,000 11,669,292 10,000,000 50,000 144,000 300,000 0 1892 1910 1930 1992 1998 2006
SLIDE 2
SLIDE 3
Sometimes, the use of trivial names is particularly convenient:
SLIDE 4
IUPAC Nomenclature
alkane: alk an e a multiplicative prefix ( # of C in parent chain) a functional group suffix a hybridization state suffix (sp3, sp2, sp)
SLIDE 5
Generating names of isomeric alkanes:
- 1. Find the longest contiguous hydrocarbon chain in the molecule. It becomes the PARENT,
- r root, name.
- 2. Identify any substituent group replacing hydrogens on the parent chain.
- 3. Name each substituent group by derivation from its parent name:
alkane - ane + yl = alkyl These alkyl groups will be used as prefixes to the parent name of the compound.
- 4. Locate all substituent group position on the parent chain by numbering the
parent chain. These numbers are called LOCANTS. When there is a choice, assign substituents the lowest possible combination of locants determined by the first point of difference in locant. Hyphens always separate locants from letters in substituent names. Substituents are listed alphabetically as prefixes to the parent name.
SLIDE 6
- 5. Identical substituents are grouped and a multiplicative prefix is used:
di, tri, tetra, penta, hexa, etc.
- 6. Complex substituents are encompassed by parentheses, or their common
names are used.
SLIDE 7
R C H H C H H C H H CH3 CH3 C C CH3 R H H H R C CH3 CH3 CH3
Simple and complex alkyl substituents classification Primary (1) Secondary (2) Tertiary (3)
SLIDE 8
IUPAC and common names of constitutional isomers of the C3 and C4 alkyl substiutents substituent common name IUPAC name n-propyl propyl isopropyl 1-methylethyl
- r
methylethyl n-butyl butyl sec-butyl s-butyl 1-methylpropyl
- r
methylpropyl isobutyl 2-methylpropyl tert-butyl t-butyl 1,1-dimethylethyl
- r
dimethylethyl
CH3 C C H R H H H R C CH3 CH3 H R C H H C H H C H H H3C CH3 C C CH3 R H H H CH3 C C H R H H3C H R C CH3 CH3 H3C
SLIDE 9
Some examples 4-(1-methylethyl)heptane 4-isopropylheptane 2,3-dimethyl-6-(2-methylpropyl)decane 6-isobutyl-2,3-dimethyldecane not 6-butyl-2,3,8-trimethylnonane not 5-isobutyl-2-isopropylnonane
SLIDE 10
H H CH3 H H CH3 H3C H H H H CH3 H3C H H H H CH3 H H3C H H H CH3 H CH3 H H H CH3 H H CH3 H H CH3 0 60 120 180 240 300 360 C1-C2--C3-C4 dihedral angle, degrees clockwise E
Conformational analysis of butane viewed along the C2-C3 sigma bond
60 120 180 240 300
SLIDE 11
H CH3 H H H3C H H H H3C H H H H3C H H H CH3 H H H H CH3 H H
0 60 120 180 240 300 360 C2-C3--C4-C5 dihedral angle, degrees clockwise E
Conformational analysis of 2,3-dimethylhexane viewed along the C3-C4 sigma bond
180 120 60 300 240
SLIDE 12
Conformations of cycloalkanes
- Baeyer observed that the planar arrays of sp3-hybridized carbons which make up the
constitutions of cylcoalkanes are generally distorted from the tetrahedral geometry: CCC = 180(n-2) ° , n where n = the number of C in the cycloalkane parent ring. CCC 60 ° 90 ° 108 ° 120 ° 129 °
- So, the relative thermodynamic stability of cycloalkanes (E / CH2) should vary as a
function of the Baeyer angle or ring strain.
SLIDE 13
SLIDE 14
Obviously, Baeyer’s predictions were not accurate! Why?
- Cycloalkanes are not necessarily flat: they may be able to make conformational
changes that can reduce ring strain.
- The thermodynamic stability of a molecule is a function of
angle strain + torsional strain + steric interactions
- These factors are not independent: changing one can increase or decrease the others
- Molecules will adjust their conformations so as to minimize strains and interactions.
- Cyclohexane appears to be able to adopt a conformation with no angle strain. Can this
conformation be determined?
SLIDE 15
Cyclohexane
All-staggered, chair conformations:
SLIDE 16
H H H H H H H H H H H H H H H H H H H H H H H H H H H H C H2 H H H C H2 H C H2 H H H H C H2 H H H H
SLIDE 17
H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H H chair chair twist-boat twist-boat boat half-chair half-chair
5.5 6.4 10.8
SLIDE 18
HH H H H H H H H H H H CH2 CH3 H H H H H H H CH2 H H CH2 H CH3 H H H CH2 H H H H H H H H H
Alkylcyclohexanes: gauche vs. anti conformations and 1,3-diaxial interactions with CH3 axial, it is gauche to CH2, but with CH3 equatorial, it is anti to CH2 another way to look at this gauche interaction: 1,3-diaxial interactions between axial substituents on the same side of the ring
SLIDE 19
O
Hydrocarbons with more than 1 ring
- Spirocycles contain 1 C common to 2 rings
spiro[4.5]decane β-vetivone
SLIDE 20
O OH OH O
Hydrocarbons with more than 1 ring
- Polycycles contain 2 C common to 2 rings
they have “shared sides” bicyclo[4.4.0] octane testosterone
SLIDE 21
O
bicyclo[3.2.1]octane camphor
SLIDE 22
Hydrocarbons with more than 1 ring
- Catenanes share no C
a [2]catenane a trefoil knot
SLIDE 23