SLIDE 1
EARLY EXAMPLES FOR RuCATALYZED LACTONIZATION O OH Ru cat. - - PowerPoint PPT Presentation
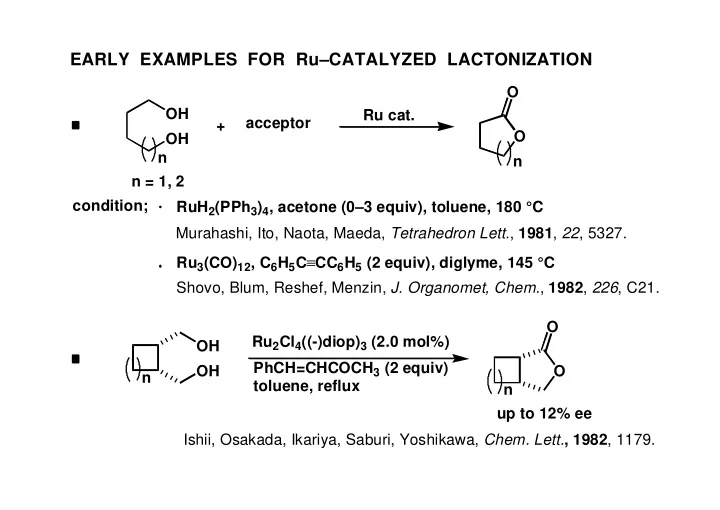
EARLY EXAMPLES FOR RuCATALYZED LACTONIZATION O OH Ru cat. - - PowerPoint PPT Presentation
EARLY EXAMPLES FOR RuCATALYZED LACTONIZATION O OH Ru cat. acceptor + O OH n n n = 1, 2 condition; RuH 2 (PPh 3 ) 4 , acetone (03 equiv), toluene, 180 C Murahashi, Ito, Naota, Maeda, Tetrahedron Lett. , 1981 , 22 , 5327. Ru 3
SLIDE 2
SLIDE 3
O R1N O R2 HP(C6H5)2 CF3SO3H i-Pr Ph Bn i-Bu R2 R1 H H H H
S
t-Bu H [CH(CH3)2] [CH2CH2(CH3)2] [C(CH3)3] [C6H5] [CH2C6H5] R1HN P(C6H5)2 R2 yield, %a 89 70 70
a Isolated yield
85
- (CH2)3-
55
SYNTHESIS OF P–N LIGANDS
66 + toluene reflux, 24 h
- xazolidinone:HP(C6H5)2:CF3SO3H = 1:2:3
S
SLIDE 4
OH OH O
ENANTIOSELECTIVE LACTONIZATION
Cp*RuCl(cod) chiral ligand KOt-Bu acetone 30 °C, 1 h R1HN P(C6H5)2 R2 H, Bn R1, R2 H, i-Pr H, i-Bu H, t-Bu H, Ph
- (CH2)3-
yield, % ee, %a diol:Ru:chiral ligand:KOt-Bu = 100:100:1:1:1 >99 >99 >99 >99 >99 >99 44 42 39 50 46 10 O
chiral ligand
S R
SLIDE 5
H2N P(C6H5)2 Bn R R OH OH R R O O
ENANTIOSELECTIVE LACTONIZATION
Cp*RuCl(cod) chiral ligand KOt-Bu 30 °C, 1 h 99% yield 2% ee 99% yield 13% ee (1S,5R) >99% yield 34% ee (1R,6S) >99% yield 31% ee (3aR,7aS) >99% yield 43% ee (2R,3S) >99% yield 50% ee (2R,3S) >99% yield 16% ee (2S,3R) >99% yield 11% ee (2S,3R) * * diol:Ru:chiral ligand:KOt-Bu = 100:100:1:1:1 O O O O O O O O O O O O O O O
chiral ligand
O
R S R S R S R S S R S R R S
SLIDE 6
OH OH O O O
CONTROL OF CONVERSION
Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOt-Bu toluene 30 °C, 1 h + diol:Ru:ligand:KOt-Bu = 100:1:1:1 diol:acetone 1 : 2 1 : 1 conv, % >99 50
SLIDE 7
H, Bn R1HN P(C6H5)2 R2
KINETIC RESOLUTION OF rac-1,4-BUTANDIOL
R1, R2 H, i-Pr H, i-Bu H, t-Bu H, Ph
- (CH2)3-
ee, %a (reactant) diol:acetone:Ru:chiral ligand:KOt-Bu = 100:100:1:1:1 79 50
chiral ligand
54 63 70 23 Ar Ar OH OH OH OH Ar Ar OH OH Ar Ar O Ar Ar O + Cp*RuCl(cod) chiral ligand KOt-Bu acetone toluene 30 °C, 1 h + 50:50 Ar = C6H5
SLIDE 8