SLIDE 1
1 / 28
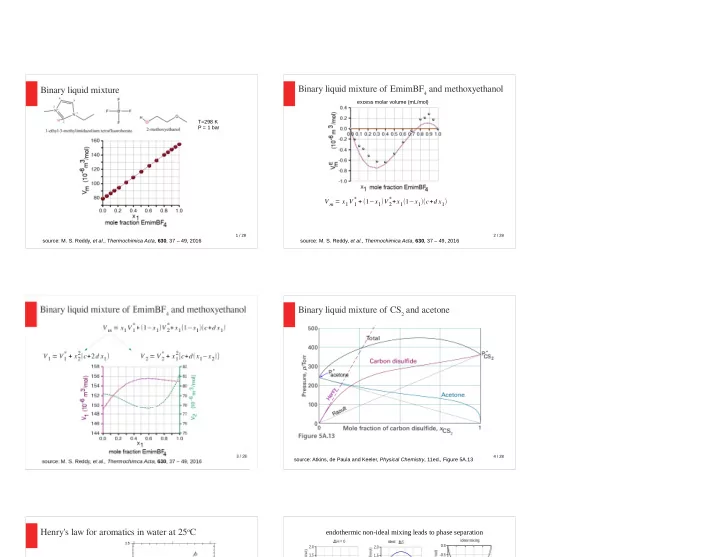
Binary liquid mixture
source: M. S. Reddy, et al., Thermochimica Acta, 630, 37 – 49, 2016 T=298 K P = 1 bar
2 / 28
Binary liquid mixture of EmimBF4 and methoxyethanol
source: M. S. Reddy, et al., Thermochimica Acta, 630, 37 – 49, 2016 excess molar volume (mL/mol)
V m = x1V1
* + (1−x1)V 2 *+x1(1−x1)(c+d x1)
4 / 28