SLIDE 1
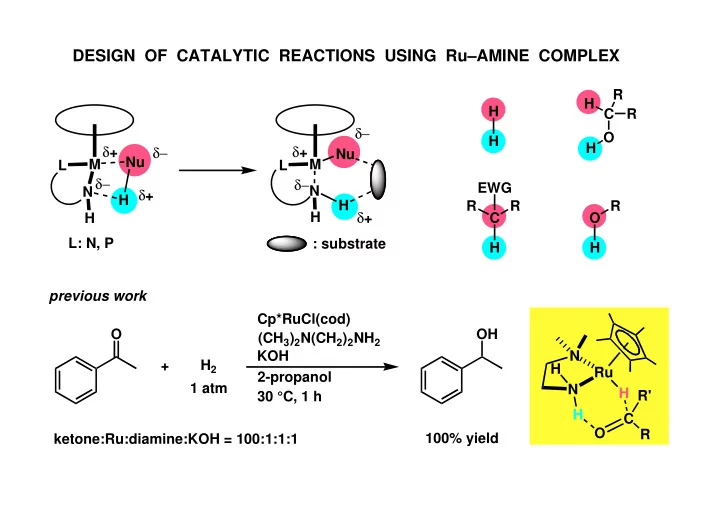
O OH 2-propanol H2 + ketone:Ru:diamine:KOH = 100:1:1:1 30 °C, 1 h 100% yield 1 atm Cp*RuCl(cod) (CH3)2N(CH2)2NH2 KOH N Ru N H H H R C R' O M N H H L Nu δ+ δ− δ− δ+ M N H H L Nu δ+ δ− δ− δ+ : substrate L: N, P
DESIGN OF CATALYTIC REACTIONS USING Ru–AMINE COMPLEX
previous work H H C O R R H H C H EWG R R O H R

SLIDE 2 Kwiatek, Mador, Seyler, J. Am. Chem. Soc., 1962, 84, 304. C6H5 C6H5 + S/C = 100, [ketone] = 0.1 M Fujitsu, Shirahama, Matumura, Takeshita, Mochida,
- J. Org. Chem., 1981, 46, 2287.
HYDROGENOLYSIS OF EPOXIDES WITH HOMOGENEOUS CATALYSTS
[Rh(nbd)(P(C2H5)3)2]+ O H2 1 atm OH C6H5 O n 90% conv + C6H5 C6H5 + S/C = 2.9, [ketone] = 0.34 M K3[Co(CN)5] O H2 1 atm OH 53% conv TOF = 0.64 h–1 diglyme–H2O 3 : 1 C6H6–H2O, r.t. 28 °C, 6 h

SLIDE 3
LIGAND ACCELERATION IN HYDROGENOLYSIS OF EPOXIDES
2-propanol H2 + Cp*RuCl(cod) ligand KOH 10 atm 30 °C, 2 h ketone:Ru:ligand:KOH = 100:1:1.5:1 OH OH branch/linear NH2 (CH3)2N (C6H5)2P N(CH3)2 NHCH3 (C6H5)2P >99/1 >99/1 (C6H5)2P P(C6H5)2 P(C6H5)3 C6H5CH2NH2 + NH2 (C6H5)2P 89/11 <1% 0% 63% conv 15% 5% <1% ligand: O +

SLIDE 4
HYDROGENOLYSIS OF TERMINAL EPOXIDES
R OH 2-propanol H2 + substrate:Ru:ligand:KOH = 100:1:1.5:1 50 °C, 18 h Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH R 100% conv + R OH 90% conv 100% conv 99% conv 99% conv 10 atm branch/linear 89/11 >99/1 >99/1 98/2 >99/1 C6H5 O O C6H5 O O O C6H5 O substrate: O intact

SLIDE 5
P Ru H N H O Ar Ar
A POSSIBLE MECHANISM
Ru HN P Ar Ar Ru H H
2
N Ar Ar P P Ru HN Ar Ar H H C6H5 O C6H5 C6H5 OH H2 Cp*RuCl(cod) KOH (C6H5)2P(CH2)2NH2 + + H

SLIDE 6
HYDROGENOLYSIS OF (S)-STYRENE OXIDE
OH 2-propanol H2 + 50 °C, 18 h Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH OH 10 atm 0% ee substrate:Ru:ligand:KOH = 100:1:1.5:1 O + 89 : 11 100% conv 96% ee

SLIDE 7
ISOTOPE LABELING EXPERIMENT
substrate:Ru:ligand:KOH = 100:1:1.5:1 2-propanol-d8 D2 + 10 atm CH2D (1.00) (0.81) CH C D2 OD (1.71) (0.93) OD D D Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH O 50 °C, 18 h R OH R O + [Ru] + R OH [Ru] + [Ru] H2 100% conv + 9 : 1

SLIDE 8
RACEMIZATION OF CHIRAL ALCOHOLS
OH 2-propanol 50 °C, 30 min Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH 0% ee 99% ee OH S/C = 100 solvent effect 2-propanol > ethanol > methanol

SLIDE 9
HYDROGENOLYSIS OF (S)-STYRENE OXIDE IN VARIOUS SOLVENTS
OH solvent H2 + 50 °C Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH OH 10 atm substrate:Ru:ligand:KOH = 100:1:1.5:1 O + 96% ee solvent conv, % branch/linear ee, % time, h 2-propanol methanol ethanol 1 2 18 79 64 64 89/11 90/10 96/4 81 84

SLIDE 10
Hydrogenation of ketones
Cp*Ru(II) COMPLEXES BEARING PRIMARY AMINE LIGANDS
Hydrogenolysis of epoxides N Ru P H H H CH2 O N Ru N H H H R C R' O R Ar Ar