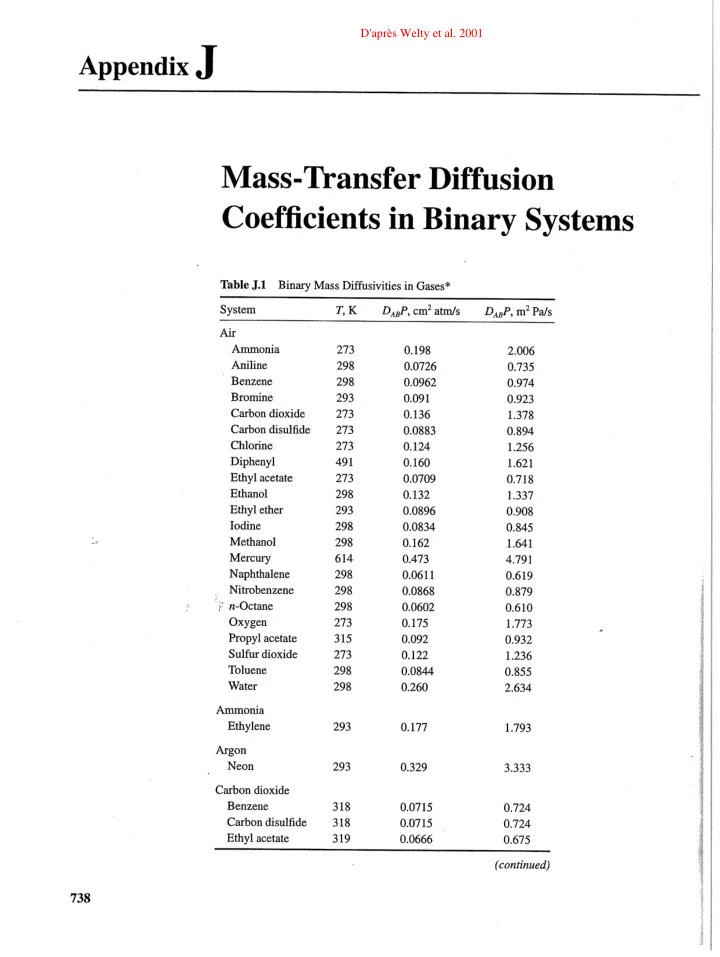
SLIDE 8 Table E.2
Collsion IntegraIs for Use with the Lennard-Jones (6-12) Potential for the
Prediction of Transport Properties of Gases at Law Densities",b,c
fll' = flk nI' = flk
KT/e
(for viscosity
fl'V,AB
KT/e
(for viscosity
n'V,AB
and thermal
(for
and thermal
(for
KT / eAB
conductivity) diffusivity)
KT / e AB
conductivity )
diffusivity)
0.30 2.840 2.649 2.7 1.0691 0.9782 0.35 2.676 2.468 2.8 1.0583 0.9682 0.40 2.531 2.314 2.9 1.0482 0.9588 0.45 2.401 2.182 3.0 1.0388 0.9500 0.50 2,284 2.066
3.1 1.0300
0.9418 0.55 2.178 1.965 3.2 1.0217 0.9340 0.60 2.084 1.877 3.3 1.0139 0.9267 0.65 1.999
3.4 1.0066 0.9197 0.70
f 1.922
1.729 3.5 0,9996 0.9131 0.75
. 1.853
1.667 3.6 0.9931 0.9068 0.80 1.790 1.612 3.7 0.9868 0.9008 0.85 1.734 1.562 3.8 0.9809 0.8952 0.90;,. 1.682 1.517 3.9 0.9753 0.8897 0.95 . 1.636 1.477 4.0 0.9699 0.8845 1.00 1.593 1.440
4.1
0.9647 0.8796 1.05 1.554
1.06
4.2 0.9598 0.8748
1.0
1.518 1.375 4.3 0.9551 0.8703
1.5
1.485 1.347 4.4 0.9506 0.8659 1.20 1.455 1.320 4.5 0.9462 0.8617 1.25 1.427 1.2?6 4.6 0.9420 0.8576 1.30 1.401 1.274 4.7 0.9380 0.8537 1.35 1.377 1.253 4.8 0.9341 0.8499 1.40 1.355 1.234 4.9 0.9304 0.8463 1.45 1.334 1.216 5.0 0.9268 0.8428 1.50 1.315
1.99
6.0 0.8962 0.8129 1.55 1.297
1.83
7.0 0.8727 0.7898 1.60 1.280
1.68
8.0 0.8538 0.7711 1.65 1.264
1.54
9.0 0.8380 0.7555 1.70 1.249
1.41
10.0 0.8244 0.7422 1.75 1.235
1.28
12.0 0.8018 0.7202 1.80 1.222
1.17
14.0 0.7836 0.7025 1.85 1.209
1.05
16.0 0.7683 0.6878 1.90 1.198 1.095 18.0 0.7552 0.6751
.,
1.95
1.86
1.085 20.0 0.7436 0.6640 2.00
1.76
1.075 25.0 0.7198 0.6414 2.10
1.56
1.058 30.0 0.7010 0.6235 2.20
1.38
1.042 35.0 0.6854 0.6088 2.30
1.22
1.027 40.0 0.6723 0.5964 2.40
1.013 50.0 0.6510 0.5763 2.50 1.0933 1.0006 75.0 0.6140 0.5415 2.60 1.0807 0.9890 100.0 0.5887 0.5180 a The values in this table, applicable for the Lennard-Jones (6-12) potential, are interpolated from the results of
- L. Monchck and E. A. Mason, ¡, Chem. Phys., 35, 1676-1697 (1961). The Monchick-Mason table is believed to be slightly
better than the earlier table by J. O. Hirschfelder, R. B. Bird, and E. L. Spotz, J. Chem. Phys., 16, 968-981 (1948).
b This table has been extended to lower temperatures by C. F. CUMS, ¡, Chem. Phys., 97, 7679-7686 (1992). Curtiss
showed that at low temperatues, the Boltzmann equation needs to be modifed to take into account "orbiting pair"
- f molecules. Only by making this modification is it possible to get a smooth transiton from quantum to classícal
- behavior. The deviations are apprecíable below dimensionless temperatures of 0.30.
C Thecollsion integrals have been curve-fitted by p, D. Neufeld, A. R. Janzen, and R. A. Aziz, ¡. Che/no Phys., 57,
1100-1102 (1972), as follows:
- = Ok = 1.6145 + 0.52487 + 2,16178
(E.2-1)
" 'T.1484 exp(O,77320T*) exp(2,43787T*)
= 1.06036 + 0.19300 + 1.03587 +
(E.2 - 2)
!1,AB T*0,1561O exp(0.47635T*) exp(1.52996T*)exp(3.89411T*)
where T* = ¡¡T / e.
866