4/29/2018 1
CHM 1045 Spring 2018 Unit I 1 4/29/2018 The process of - - PDF document
CHM 1045 Spring 2018 Unit I 1 4/29/2018 The process of - - PDF document
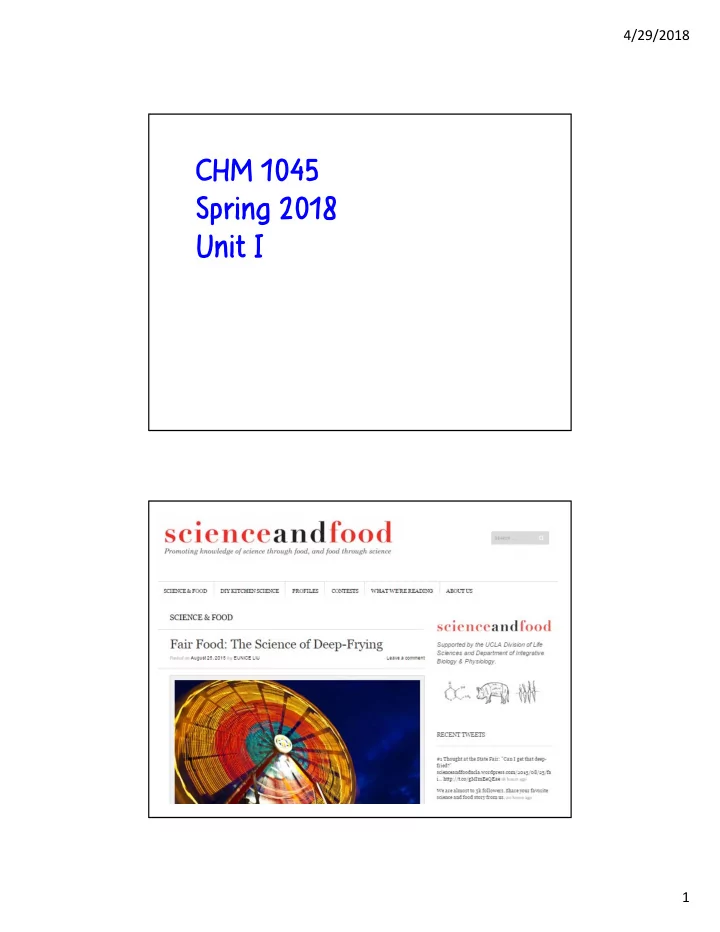
4/29/2018 CHM 1045 Spring 2018 Unit I 1 4/29/2018 The process of deep-frying can be divided into four stages: (1) Initial heating. The surface of the food reaches the boiling point of water. (2) Surface boiling. The hot oil surrounding the
4/29/2018 2
The process of deep-frying can be divided into four stages:
(1) Initial heating. The surface of the food reaches the boiling point of water. (2) Surface boiling. The hot oil surrounding the food causes water inside the food to evaporate. (3) Decreasing heat transfer rate. Crust continues to dehydrate, less heat conduction, rate of heat transfer through escaping steam decreases (reduced bubbling). The remaining moisture inside of the food is slowly heated to the boiling point of water, which cooks the food inside as if it were boiled, gelatinizing the starch and denaturing the proteins in the food. (4) Bubble end point. Water from inside the food is no longer evaporating. Remove the fried food before it begins to absorb oil.
https://scienceandfooducla.wordpress.com/2015/08/25/fair-food-deep-frying/
4/29/2018 3
Indiana Jones and the Raiders of the Lost Arc
Paramount, 1981
Assuming the statue is solid gold, how much will it weigh?
- A. 10 lb
- B. 20 lb
- C. 40 lb
- D. 80 lb
- E. 160 lb
4/29/2018 4
Assuming the statue is solid gold, how much will it weigh?
- A. 10 lb
- B. 20 lb
- C. 40 lb
- D. 80 lb
- E. 160 lb
4/29/2018 5
Assuming the statue is solid gold, how much will it weigh?
- A. 10 lb
- B. 20 lb
- C. 40 lb
- D. 80 lb
- E. 160 lb
4/29/2018 6
René Descartes (1596-1650)
4/29/2018 7
Daniel Gabriel Fahrenheit German physicist 1709, alcohol thermometer 1714, mercury thermometer 1724, Fahrenheit Scale, allowed temperatures to be recorded reproducibly. Anders Celsius Swedish astronomer 1742, devised the centigrade
- r "Celsius scale"
4/29/2018 8
0 K
900 megahertz, 21.1-tesla NMR magnet at the FSU National High Magnetic Field Laboratory (Mag Lab)
liquid helium
4.2 K
4/29/2018 9
How much does it cost to drive from FSU (Tallahassee) to UF (Gainesville)?
4/29/2018 10
4/29/2018 11
4/29/2018 12
(4.8 x 10-19 C)/(1.6 x 10-19 C) = 3 3 e (3.2 x 10-19 C)/(1.6 x 10-19 C) = 2 2 e (6.4 x 10-19 C)/(1.6 x 10-19 C) = 4 4 e (1.6 x 10-19 C)/(1.6 x 10-19 C) = 1 1 e (4.8 x 10-19 C)/(1.6 x 10-19 C) = 3 3 e
Assuming the lowest charge corresponds to having only one electron transferred to the oil drop, implies....
4/29/2018 13
http://www.school-for-champions.com/science/static_materials.htm
Becom e positive in charge
The following m aterials will tend to give up electrons when brought in contact with- ther materials. They are listed from those with the greatest tendency to give
Neutral
There are very few m aterials that do not tend to readily attract or give up electrons when brought in contact or rubbed with other m aterials. Cotton Best for non-static clothes Steel Not useful for static electricityBecom e negative in charge
The following materials will tend to attract electrons when brought in contact with- ther materials. They are listed from those with the least tendency to attract
Triboelectric Series
An effort to reconstruct Millikan's "exemplary" experimental thinking revealed serious discrepancies between Millikan's notebooks and his published "raw" data (Holton, 1978). The numerous notes which are scattered across the pages cast further doubt
- n Millikan's integrity:
This is almost exactly right & the best one I ever had!!! [20 December 1911] Exactly right [3 February 1912] Publish this Beautiful one [24 February 1912] Publish this surely / Beautiful !! [15 March 1912, #1] Error high will not use [15 March 1912, #2] Perfect Publish [11 April 1912] Won't work [16 April 1912, #2] Too high by 1½% [16 April 1912, #3] http://www1.umn.edu/ships/ethics/millikan.htm
4/29/2018 14
The notebooks reveal that, indeed, substantial data are missing from Millikan's published reports. Of 175 total drops documented in the notebooks, only 58 (barely one-third) appear in the final paper. By contrast, Millikan had announced in his 1913 paper that "It is to be remarked, too, that this is not a selected group
- f drops but represents all of the drops experimented on during 60 consecutive
days, during which time the apparatus was taken down several times and set up anew" [his own emphasis!]. In his 1917 book, The Electron, he repeats this statement and then adds, "These drops represent all of those studied for 60 consecutive days, no single drop being omitted.“ At first blush, this outrageous violation of scientific integrity would seem to discredit Millikan's findings. Even if one assumes that standards of reporting data earlier in the century were less rigorous, Millikan clearly misrepresented the extent of his data. One may caution students, however, that we may not want to conclude that therefore there was no good, "scientific" basis for his selective use
- f data. A more complete analysis of Millikan's notebooks, in fact, and of the
nature of the experimental task that they crudely document, reveals more tellingly the reasons that Millikan included some drops and excluded others. http://www1.umn.edu/ships/ethics/millikan.htm
4/29/2018 15
4/29/2018 16
It the nucleus was the size
- f a soda
bottle cap.... ... a typical atom would be this big!
4/29/2018 17
- A. S
- B. Se
- C. Se2
- D. Br
- E. Br2
Typical question, but a more realistic question would be... What is the formula of the compound you would expect to form between potassium and selenium, and how would you name that compound?
4/29/2018 18
The ionic compound formed between calcium and phosphorous is used to make incendiary bombs and also as a rodenticide. What is the expected formula for this compound?
- A. CaP
- B. CaP2
- C. Ca2P
- D. Ca2P3
- E. Ca3P2
What is the name of the compound Rb2S?
- A. rubidium sulfide
- B. dirubidium sulfide
- C. dirubidium monosulfide
- D. rubidium(I) sulfide
- E. dirubidium(I) sulfide
4/29/2018 19
https://en.wikipedia.org/wiki/Nitrogen_oxide
4/29/2018 20
- ne gross lightbulbs = 144 lightbulbs
- ne dozen donuts = 12 donuts
- ne ream paper = 500 sheets of paper
- ne mole water molecules =
602,200,000,000 000,000,000,000 water molecules
You are throwing a party for 12 people. Assume each person will eat 9 shrimp. How many pounds of “18-count” shrimp should you buy?
- A. 3 lbs
- B. 6 lbs
- C. 9 lbs
- D. 12 lbs
- E. 15 lbs
4/29/2018 21
mass of H2O absorbed moles of H2O absorbed moles of H in sample mass of CO2 absorbed moles of CO2 absorbed moles of C in sample
4/29/2018 22
4/29/2018 23
4/29/2018 24
What is the name
- f the compound Fe2O3?
4/29/2018 25
The following slides are from when I used to use clickers in this class. We asked a few of these in class, but I included the original versions here for additional practice. Iodine-131 is used to image the brain and monitor thyroid activity, but it is also a byproduct of nuclear fusion and causes cancer. An 131I nucleus contains: A. 53 protons and 78 neutrons B. 53 protons and 131 neutrons C. 78 protons and 53 neutrons D. 131 protons and 53 neutrons E. 131 protons and 78 neutrons
4/29/2018 26
Iodine-131 is used to image the brain and monitor thyroid activity, but it is also a byproduct of nuclear fusion and causes cancer. An 131I nucleus contains: A. 53 protons and 78 neutrons B. 53 protons and 131 neutrons C. 78 protons and 53 neutrons D. 131 protons and 53 neutrons E. 131 protons and 78 neutrons What is the name of the compound Rb2S?
- A. rubidium sulfide
- B. dirubidium sulfide
- C. dirubidium monosulfide
- D. rubidium(I) sulfide
- E. dirubidium(I) sulfide
4/29/2018 27
What is the name of the compound Rb2S?
- A. rubidium sulfide
- B. dirubidium sulfide
- C. dirubidium monosulfide
- D. rubidium(I) sulfide
- E. dirubidium(I) sulfide
The ionic compound formed between calcium and phosphorous is used to make incendiary bombs and also as a rodenticide. What is the expected formula for this compound?
- A. CaP
- B. CaP2
- C. Ca2P
- D. Ca2P3
- E. Ca3P2
4/29/2018 28
The ionic compound formed between calcium and phosphorous is used to make incendiary bombs and also as a rodenticide. What is the expected formula for this compound?
- A. CaP
- B. CaP2
- C. Ca2P
- D. Ca2P3
- E. Ca3P2
What is the name of this compound?
What is the name of the compound PCl3?
- A. phosphorus chloride
- B. phosphorus(III) chloride
- C. phosphorus trichloride
- D. monophosphorus trichloride
- E. phosphorus(III) trichloride
4/29/2018 29
What is the name of the compound PCl3?
- A. phosphorus chloride
- B. phosphorus(III) chloride
- C. phosphorus trichloride
- D. monophosphorus trichloride
- E. phosphorus(III) trichloride
What is the name of the compound Fe(NO2)3?
- A. iron trinitrite
- B. iron(II) nitrite
- C. iron(III) nitrite
- D. iron trinitrate
- E. iron(III) nitrate
4/29/2018 30
What is the name of the compound Fe(NO2)3?
- A. iron trinitrite
- B. iron(II) nitrite
- C. iron(III) nitrite
- D. iron trinitrate
- E. iron(III) nitrate
1
CHM 1045 Spring 2018 Unit II
In Section 4.1 we learned how to balance this reaction:
Pb(NO3)2(aq) + KI(aq) PbI2(s) + KNO3(aq)
2
In Section 4.2, we learn how to balance this reaction:
Pb(NO3)2(aq) + KI(aq)
In Chapter 4, we learn how to balance this reaction:
Pb(NO3)2(aq) + KI(aq)
3
page 188
The following guidelines are used to assign
- xidation numbers to each element in a molecule or ion.
- 1. The oxidation number of an atom in an elemental substance is zero.
- 2. The oxidation number of a monatomic ion is equal to the ion’s charge.
- 3. Oxidation numbers for common nonmetals are usually assigned as follows:
- Hydrogen: +1 when combined with nonmetals, −1 when combined with metals
- Oxygen: −2 in most compounds, sometimes −1 (so-called peroxides, O2
2−),
very rarely − 1/ 2 (so-called superoxides, O2
−), positive values when combined
with F (values vary)
- Halogens: −1 for F always, −1 for other halogens except when combined with
- xygen or other halogens (positive oxidation numbers in these cases, varying
values)
- 4. The sum of oxidation numbers for all atoms in a molecule or polyatomic ion
equals the charge on the molecule or ion.
4
page 188
5
starts at g ethanol__ 100 mL blood
impaired
0.04
illegal (DUI)
0.08
intoxicated (drunk)
0.16
lethal
0.4 American Medical Association Guidelines
6
3 2 3 2 3 2
0.0485 mol CH CH OH 46.07 g CH CH OH 1000 mL x x L mol CH CH OH 1 L x 100 mL plasma = 0.223 g ethanol per 100 mL blood plasma
7
http://www.breathalyzerforsale.net/bactrack-element.html
8
The Blindside, 2009
9
Minor facts were changed. The Tuohy’s daughter, Collins, actually pole vaulted instead of playing volleyball. She was the same grade as Michael. The car accident was on a different street. But the scene where Michael Oher stops an air bag from hitting their son wasn’t embellished. "That’s 100 percent accurate,” Sean said. "The car behind them was someone from the basketball team. Someone witnessed that. The overall theme and concept was dead on, more accurate than Hollywood usually does.”
Read more: http://newsok.com/sean-tuohy-stunned-by- fame/article/3424145#ixzz28hZWEOrU
Chapter 9 – Gases
The Ideal Gas Law Pressure and Named Gas Laws Non-ideal Gases Kinetic- Molecular Physics
PV = nRT
10
3 3 2
453.6 g 14.7 lb 1 cm 1 in 1 lb 7.85 g 2.54 cm 1 in
Steel “Atmosphere Bar”
51.8 in 4 ft, 3.8 in
Aluminum Lead 3 ft Osmium 1.5 ft ($670,000) 12 ft, 6.6 in
11
h = 24.0 cm, so the pressure
- f the gas is...
Given that the atmospheric pressure is 753 mmHg and h = 14.1 cm, the pressure of the gas is...
12
0 K
Recall...
Teams of horses cannot pull apart pull apart “vacuum- filled” copper spheres in Magdeburg demonstration for Emperor Ferdinand III in 1654.
Otto von Guericke 1602-1686
13
0 psi-g “gauge pressure” is actually 14.7 psi
Automobile tires at 35 psi are really at 35 psi-g (psi “gauge”). The absolute pressure would be 35 + 15 = 50 psi. A vacuum is 14.7 psi-g.
14
15
16
1 2 1 2 1 2 i f 1 2 i f
PV = nRT P P P nR nR assuming V is constant => = => = = T V T V T P P P P So =
- r
= T T T T
i f f i
P T 27.2 psi x 282 K P = = = 25.9 psi T 296 K Ti = 23 oC + 273.15 = 296 K Tf = 9 oC + 273.15 = 282 K Rearranging this expression, the final pressure at halftime would be
.
25.9 psi (absolute) - 14.7 = 11.2 psi-g Below the 12.5 - 13.5 “psi” required by the NFL, but not 2 psi below the minimum allowed pressure
17
18
The pressure on a diver increases by 100 kPa (1.00 atm) for every 10 m the diver descends. For dives deeper than 66 m the gas mixture should contain less than 21%
- xygen to avoid the risk of acute oxygen toxicity. The general rule is to try to
achieve a gas mixture giving an Fio2 of about 140 kPa. At 130 m depth in the northern sector of the North Sea oil field, the ambient pressure is 1400 kPa, so the breathing mixture used contains 10% oxygen. On the deepest working dives, at depths greater than 600 m, ambient pressure is greater than 6100 kPa and the divers breathe gas mixtures containing about 2% oxygen to avoid acute oxygen toxicity. A lung full of gas containing 2% oxygen at 600 m contains about six times as many molecules of oxygen as a lung full of air at sea level. On deep dives the composition of the gas breathed is changed several times during descent and ascent. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1114047/ 600 m => 6100 kP => 60 atm! 60 x (2%/100%) => P(O2) = 1.2 atm
https://en.wikipedia.org/wiki/Partial_pressure#In_medicine
19
http://slideplayer.com/slide/4772848/
20
21
A gaseous compound is 30.4% nitrogen and 69.6% oxygen by mass. A 5.25-g sample of the gas occupies a volume of 1.00 L and exerts a pressure of 1.26 atm at 4.0°C. What is the molecular formula of this compound? A gaseous compound is 30.4% nitrogen and 69.6% oxygen by mass. A 5.25-g sample of the gas occupies a volume of 1.00 L and exerts a pressure of 1.26 atm at 4.0°C. What is the molecular formula of this compound? A. NO2 B. N2O4 C. N3O6 D. N4O8
22
An ideal gas is trapped in a flexible container – perhaps a massless, frictionless piston. The pressure is increased by a factor of two and the temperature is decreased by a factor of two. The volume will…
- A. quadruple.
- B. double.
- C. remain the same.
- D. decrease to 1/2 original.
- E. decrease to 1/4 original.
2NaN3(s) 2Na(s) + 3N2(g)
https://www.youtube.com/watch?v=IzC_QqKhTQg
23
24
http://www.ncert.nic.in/html/learning_basket/energy10class/combustion%20engine1.htm http://www.ncert.nic.in/html/learning_basket/energy10class/combustion%20engine1.htm
25
How many servings of CoolWhip in an 8 oz container?
- A. less than 10
- B. between 10 and 20
- C. between 20 and 30
- D. between 30 and 40
- E. greater then 40
chemical energy (fuel) you add energy you release (light, heat, motion)
fuel + O2 CO2 + H2O
26
How much does it cost to drive from FSU (Tallahassee) to UF (Gainesville)?
$3.50 = $42 1 gal 2 150 mi 25 mi 1 gal 2 1 ga 150 mi 2 l 5 mi
....
27
28
chemical energy (fuel) you add energy you release (light, heat, motion)
fuel + O2 CO2 + H2O food + O2 CO2 + H2O
chemical energy (food) you add energy you release (heat, to maintain body temperature; motion, to do work)
29
30
31
We use stored energy from food at a rate ranging from about 1 Cal/min (basal metabolic rate) to about 15 Cal/min.
32
Assumptions: No exercise beyond the normal activity to burn 2000 Calories per day. No food. Get energy from glycogen until gone, then burn fat after that.
http://www.uic.edu/classes/phar/phar332/Clinical_Cases/carbo%20metab%20cases/ glycogen%20metab/Glycogen%20biochemistry.htm
33
Assuming an energy need of 2000 Cal/day and no food (!) during this time: 2000 Cal/day (1 g/9 Cal) (1 lb/453.6 g) 0.4899 lb/day So, max weight loss in 20 days is 1 lb (1st day, glycogen) + 19 day(0.5 lb/day) = 10.5 lb !
34
H2 O2 2:1 H2+O2 Which balloon contains the most molecules?
- A. the H2 balloon
- B. the O2 balloon
- C. the H2/O2 balloon
- D. they all contain the same number of
molecules
35
H2 O2 2:1 H2+O2 Which balloon is the heaviest?
- A. the H2 balloon
- B. the O2 balloon
- C. the H2/O2 balloon
- D. all three balloons are equally heavy
H2 O2 2:1 H2+O2 Which balloon is the densest?
- A. the H2 balloon
- B. the O2 balloon
- C. the H2/O2 balloon
- D. all three balloons are equally dense
36
H2 O2 2:1 H2+O2 Which balloon released the most energy?
- A. the H2 balloon
- B. the O2 balloon
- C. the H2/O2 balloon
1
http://www.uic.edu/classes/phar/phar332/Clinical_Cases/carbo%20metab%20cases/ glycogen%20metab/Glycogen%20biochemistry.htm
CHM 1045 Spring 2018 Unit III
2
http://www.uic.edu/classes/phar/phar332/Clinical_Cases/carbo%20metab%20cases/ glycogen%20metab/Glycogen%20biochemistry.htm
NH4NO3(s) NH4
+(aq) + NO3 (aq)
3
Oil of wintergreen contains methyl salicylate which has anti-inflammatory properties and is closely related to the well-known medication aspirin
Wintergreen
H O O O
aspirin
(acetylsalicylic acid)
4
Bob Holton
Department of Chemistry and Biochemistry Florida State University
Total Synthesis –
Synthesis of a “natural product” from all non-natural (i.e. not biological in origin) starting materials; yields often very low. So why do it? It’s hard; it’s a race (i.e. there is a winner); the synthetic procedure you develop to solve this problem can then be applied to other desired compounds; the students who do this work go on to solve other important problems.
“Semi”-synthesis –
Synthesis of a “natural product” starting with a readily available natural product of lesser complexity; hence much higher yields. Why do it? More people get the drug; oh yes..... $$$$$$$
5
Holton started with 10- Deacetylbaccatin III (or 10- DAB), a natural compound found in abundance in the needles of the English yew, a common European
- shrub. Attaching a 34 atom
ester side-chain at a particular site, plus a few
- ther steps, produced
significant amounts of Taxol.
6
7
Orbitals (shapes and energies) Quantum numbers (n, l, ml)
Quantum (wave) mechanics
Dual particle- wave nature
- f matter
Electro- magnetic radiation Waves
Retrosynthesis
- f Atomic
Orbitals
Why are fire trucks red?
8
Why are fire trucks red?
- A. Red paint is the cheapest.
- B. People like red the most.
- C. Red paint is easiest to see.
In the early 1970s, Ward LaFrance created what was probably the most controversial issue ever to hit the apparatus field. By aggressively marketing a new color for fire apparatus, lime green, as being more visible and therefore safer, a wedge was driven into the fire service. Traditionalists stood behind the old standard red, while "progressive" fire service personnel preached the advantages of the new color.
9
At one point, there were actually more apparatus being delivered in the new color than the traditional red. Many fire departments, both large and small, switched to the new color.
http://www.firehouse.com/magazine/american/apparatus.html
Contemporary Color Theory and Use: Theory and Use Steven Bleicher, Cengage Learning, 2004; page 42
For years, fire trucks have been painted red, hence the name “fire-truck red.” But several years ago it was discovered that this red hue is hard to see at dawn and dusk when there is little
- light. Today, yellow-green is used for fire trucks
and hydrants because it can be seen from farthest awar, regardless of the light and time
- f day. Years from now when school children ask
what color fire trucks are, they will answer “yellow-green.”
10
http://acept.asu.edu/PiN/rdg/rainbow/secondary.shtml FSU vs. NCSU game October 6, 2007
11
12
= wavelength = frequency c = speed of light
c =
1/
1
13
http://www.smc.edu/AcademicPrograms/PhysicalSciences/Documents/ Chemistry_11_Experiments/Mn_Lab.pdf
ROY G. BIV
14
15
= wavelength = frequency c = speed of light
c =
1/
=========================================
blackbody radiation, photoelectric effect
E = Ephoton = h
1
http://webvision.med.utah.edu/imageswv/spectra.jpeg
16
http://www.amastro2.org/at/ot/othcs.gif
near-IR
17
Why are fire trucks red?
- A. Red paint is the cheapest.
- B. People like red the most.
- C. Red paint is easiest to see.
Many fire departments, both large and small, switched to the new color. But after about a decade, departments began to switch back to red. Many apparatus were repainted. Virtually every large department that had gone to the new color returned to red. Currently, few new apparatus are being delivered that are painted lime green.
http://www.firehouse.com/magazine/american/apparatus.html
18
“the size of the wave matches the size of the antenna”
processes involving the nucleus processes involving core electrons excite/remove valence and bonding electrons vibrate bonds between atoms rotate molecules 20 cm = 0.020 m
19
What wavelength of light would be required to break a carbon-chlorine bond?
20
21
electrons diffracted off aluminum foil X-rays diffracted off aluminum foil
22
1 wavelength
23
24
De Broglie wavelength = h/(mv)
everything has a wavelength, but it only becomes important at very low masses (electrons, atoms) or very low speeds
25
Deion “Primetime” Sanders – sports analyst, recording artist, professional athlete in two sports, and star FSU athlete in three sports – ran a 4.27 second 40-yard dash in 1989, within 0.15 s of the current world record. Determine Deion Sanders’ wavelength when he ran the 40-yard dash in 4.27 s. (Note: 1 J = 1 kg∙m2/s2)
De Broglie wavelength = h/(mv)
everything has a wavelength, but it only becomes important at very low masses (electrons, atoms) or very low speeds
Heisenberg uncertainty principle x p ≥ h/(4)
- 1. we cannot know anything with perfect accuracy; i.e. no determinism
- 2. the very act of measuring something changes the thing we were
measuring; i.e. there are no innocent measurements – we are tied to what we observe
- 3. at the atomic level, the universe is Bizarro World
http://www.youtube.com/watch?v=DfPeprQ7oGc
26
n principal quantum number n can have values 1, 2, … gives info about energy and distance from nucleus
l
- rbital angular momentum quantum number
l can have values 0 (s), 1 (p), 2 (d), 3 (f), .... n-1
gives information about shape
s orbital p orbitals d orbitals
ml magnetic quantum number ml can have the values (l), (l)+1, ... 0, 1 ..., l1, l gives the number of “orbitals” (2l + 1) for that l value
27
Energy n = 1 n = 2 n = 3 n = ∞ E1 = -2.18 x 10-18 J E2 = -5.45 x 10-19 J E3 = -2.42 x 10-19 J E∞ = 0 J … …
En = -h2/(82meao
2n2) = -2.18 x 10-18 J/n2
How many n = 4 orbitals are possible for an atom? A. 4 B. 8 C. 16 D. 32 E.
- ne 4s orbital
three 4p orbitals five 4d orbitals seven 4f orbitals 1 + 3 + 5 + 7 = 16 orbitals
28
How many l = 2 orbitals (i.e. d orbitals) are possible for an atom? A. B. 1 C. 3 D. 5 E.
five 3d orbitals five 4d orbitals five 5d orbitals five 100d orbitals etc...
How many 2d orbitals are possible for an atom? A. B. 1 C. 3 D. 5 E.
d => l = 2 => n may be 3, 4, ... So there are no 2d orbitals
29
How many 3d orbitals are possible for an atom? A. B. 1 C. 3 D. 5 E. How many 4f orbitals are possible for an atom? A. 5 B. 7 C. 10 D. 12 E. 14
30
1 e system (H, He+, Li2+…) multi-electron ( 2e) system
31
32
The rest of the story, according to Stern: Physics Today, December 2003
1
CHM 1045 Spring 2018 Unit IV
Which one of these atoms is the smallest?
- A. P
- B. Cl
- C. Sb
- D. I
2
Which one of these atoms has the greatest ionization energy? In other words, which
- ne is hardest to ionize?
- A. P
- B. Cl
- C. Sb
- D. I
3
4
fill ns orbitals 1s, 2s, 3s, 4s... fill np orbitals 2p, 3p, 4p... fill nd orbitals 3d, 4d, 5d
BE (kJ/mol) bond length (pm) F2 159 143 O2 498 121 N2 946 110
5
?
6
Which is the best dot structure for CO2? B. A.
7
Which is the best dot structure for CO2?
O C O O C O
+ _
A. B.
8
9
Increasing electronegativity Special cases: (1) EN(O) > EN(Cl); (2) EN(H) EN(C).
10
A triatomic compound contains one nitrogen atom, one
- xygen atom, and one fluorine atom. What is the most
likely arrangement of the atoms?
O N F O F N N O F
2+ _ _ + _
O N F O F N N O F
2+ _ + + _ 2 _ 2 +
What is the most likely arrangement of the atoms? A) B) C)
11
12
13
Chiral molecules have 4 different groups attached to a central atom. A chiral molecule and its non-superimposable mirror image are a special kind of isomer called enantiomers. Enantiomers have identical physical properties. The only way we can tell them apart is by their effect
- n plane polarized light.
14
15
16
Aspartame Dulcin Sodium Cyclamate Saccharine Sucralose
very similar
???
17
50% 50% laboratory synthesis
18
19
Our final exam will be administered in 275 FLH (not 255 FLH, our usual classroom) and it will be roughly half new material since Exam 3 and half cumulative covering the material up to and through Exam 3. The final exams in general chemistry are given as block exams. Our final exam is Monday, April 30, from 12:30 pm to 2:30 pm.
20
Lone pairs + atoms = 5 Arrangement is trigonal bipyrimidal 0 lone pairs molecular geometry is trigonal bipyrimidal 1 lone pair molecular geometry is… 2 lone pairs molecular geometry is…
21
90° lp/lp: 90° lp/bp: 90° bp/bp:
2 lone pairs molecular geometry is…
A. B. C. 1 lone pair molecular geometry is see-saw 2 lone pairs molecular geometry is T-shaped (or bent T) 3 lone pairs molecular geometry is linear
B A B B B
22
lone pairs + atoms = 6 arrangement is octahedral 0 lone pairs; molecular geometry is
- ctahedral
1 lone pair; molecular geometry is square pyramidal 2 lone pairs; molecular geometry is square planar
23
Give the molecular shape of the following molecules: SO2 SO3 A. linear trigonal planar B. linear trigonal pyramidal C. bent trigonal planar D. bent trigonal pyramidal
24
Are SO2 and SO3 polar or nonpolar? SO2 SO3 A. polar polar B. nonpolar polar C. polar nonpolar D. nonpolar nonpolar
http://chemwiki.ucdavis.edu/Wikitexts/UCD_Chem_2A/ChemWiki _Module_Topics/Chemical_Bonding/Electrostatic_Potential_maps
25
Everything we learned this semester is wrong...
(Slides from the last day of class. Hybridization and molecular orbital theory will not be on our exam.)
26
27
Hybridization: sp sp2 sp3 sp3d sp3d2