11/13/2013 1
CEE 697K
ENVIRONMENTAL REACTION KINETICS
Introduction
David A. Reckhow
CEE 679 Kinetics Lecture #18 1
Updated: 13 November 2013
Print version
Lecture #18
Chloramines with Surface Reactions: Pipe walls & degradation in Distribution Systems Primary Literature
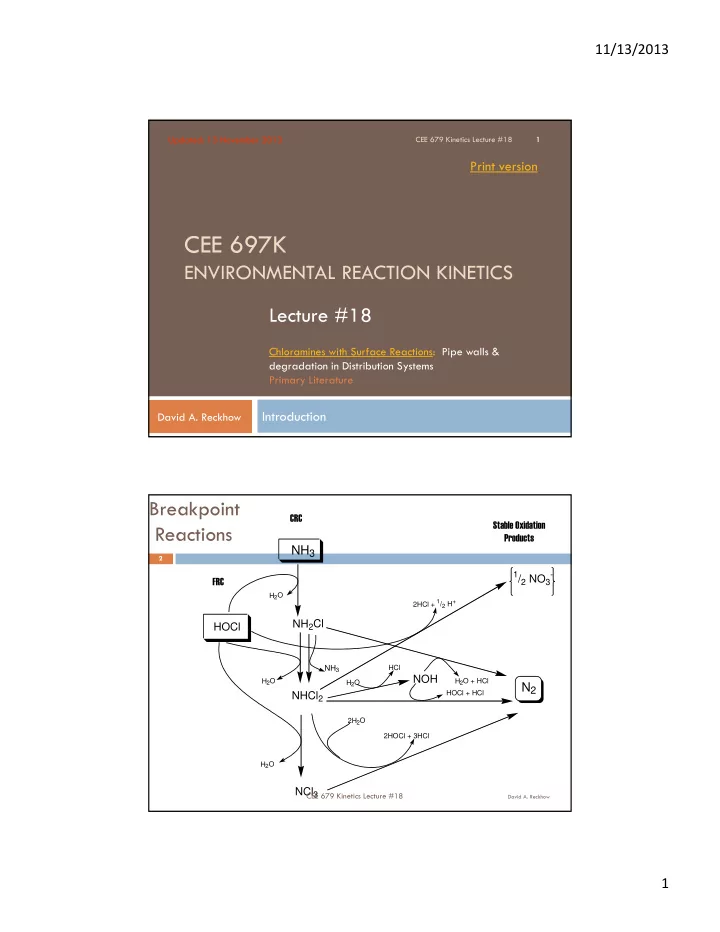
H2O H2O
NH3
NCl3 NH2Cl HOCl NOH NHCl2
H2O H2O 2H2O
1/2 NO3
- 2HCl + 1/2 H+
N2
H2O + HCl HOCl + HCl HCl 2HOCl + 3HCl
NH3
Breakpoint Reactions
Stable Oxidation Products FRC CRC
David A. Reckhow
CEE 679 Kinetics Lecture #18
2