SLIDE 1
CH BOND ACTIVATION WITH METAL AMIDE COMPLEXES Ts Ts R R N - - PowerPoint PPT Presentation
CH BOND ACTIVATION WITH METAL AMIDE COMPLEXES Ts Ts R R N - - PowerPoint PPT Presentation
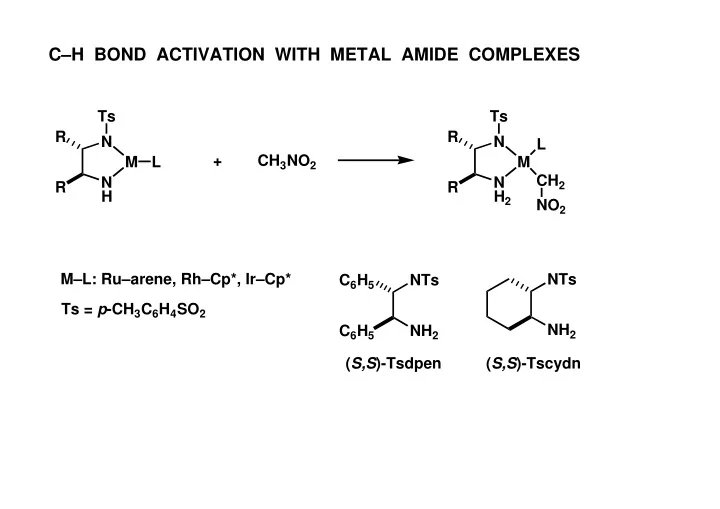
CH BOND ACTIVATION WITH METAL AMIDE COMPLEXES Ts Ts R R N N L + CH 3 NO 2 M L M CH 2 N N R R H H 2 NO 2 ML: Ruarene, RhCp*, IrCp* NTs C 6 H 5 NTs Ts = p -CH 3 C 6 H 4 SO 2 NH 2 C 6 H 5 NH 2 ( S,S )-Tsdpen (
SLIDE 2
SLIDE 3
CH3NO2 + 2-methyl-2-butanol 30 ˚C, 24 h H O OH NO2
S
chiral cat 37 40 ee, % 29 25 chiral cat
NITROALDOL REACTION WITH CHIRAL METAL COMPLEXES
Ru[(S,S)-Tsdpen](p-cymene) Cp*Rh(CH2NO2)[(S,S)-Tscydn] Cp*Ir(CH2NO2)[(S,S)-Tscydn] yield, % Conditions: cat:aldehyde:CH3NO2 = 1:50,100, [aldehyde] = 0.2 M
SLIDE 4
CH3NO2 + chiral Ru cat 24 h, 30 ˚C C6H5 H O C6H5 OH NO2
S
conv, % solvent 88 40 yield, % 37 ee, % 2-methyl-2-butanol 80 30 30 2-propanol 34 30 10 toluene 6 39 4 THF
SOLVENT EFFECT FOR ASYMMETRIC NITROALDOL REACTION
Conditions: Ru:aldehyde:CH3NO2 = 1:50:100, [aldehyde] = 0.2 M methanol 100 13 17 solvent chiral Ru cat: Ru[(S,S)-Tsdpen](p-cymene)
SLIDE 5
CH3NO2, equiv temp, ˚C 2 30 88 40 50 33 conv, % 45 2 ee, %
EFFECTS OF THE AMOUNT OF CH3NO2
5 10 36 63 5 30 10 30 26 7 81 100 yield, % 32 37 56 65 83 C6H5 OH NO2
S
Conditions: Ru:aldehyde = 1:50, [aldehyde] = 0.2 M in 2-methyl-2-butanol chiral Ru cat: Ru[(S,S)-Tsdpen](p-cymene)
SLIDE 6
10 20 80 100 conv, % ee, % 60 40 20 30 80 100 60 40 20 time, h conv, % ee, % chiral Ru cat: Ru[(S,S)-Tsdpen](p-cymene)
TIME DEPENDENCE OF EE VALUES
Condition: Ru:aldehyde:CH3NO2 =1:50:100, [aldehyde] = 0.2 M in 2-methyl-2-butanol, 30 ˚C
SLIDE 7
chiral Ru cat 24 h R OH NO2
S
R H O 2-methyl-2-butanol
ASYMMETRIC NITROALDOL REACTION OF ALDEHYDES
R C6H5(CH2)2 C6H5 C6H11 yield, % ee, % 56 36 20 20 18 75 S/C = 50 temp, °C CH3NO2 + CH3NO2, equiv 10 30 30 5 2 2 Conditions: [aldehyde] = 0.2 M chiral Ru cat: Ru[(S,S)-Tsdpen](p-cymene)
SLIDE 8
REACTION OF PHENYLPROPANAL WITH NITROETHANE
+ CH3CH2NO2 chiral Ru cat NO2 OH NO2 OH Ru:aldehyde:nitroethane = 1:50:100, [aldehyde] = 0.2 M + C6H5 H O C6H5 C6H5 30 ˚C, 24 h 2-methyl-2-butanol 48% yield syn anti 1:1
S S
51% ee
S R
33% ee chiral Ru cat: Ru[(S,S)-Tsdpen](p-cymene)
SLIDE 9
chiral Ru cat R OH NO2 R H O 2-methyl-2-butanol
CONCLUSION
R'CH2NO2 + 36–75% ee 18–56% yield S/C = 50 chiral Ru cat: Ru[(S,S)-Tsdpen](p-cymene) 10–30 °C, 24 h R'CH2NO2: CH3NO2, CH3CH2NO2 R' * *
ACKNOWLEDGEMENTS
- Prof. M. Shibasaki (Tokyo Univ. )