Bonding Bonding: H2+ and H2 molecules ( h h h ) 2 2 2 + = - - PowerPoint PPT Presentation
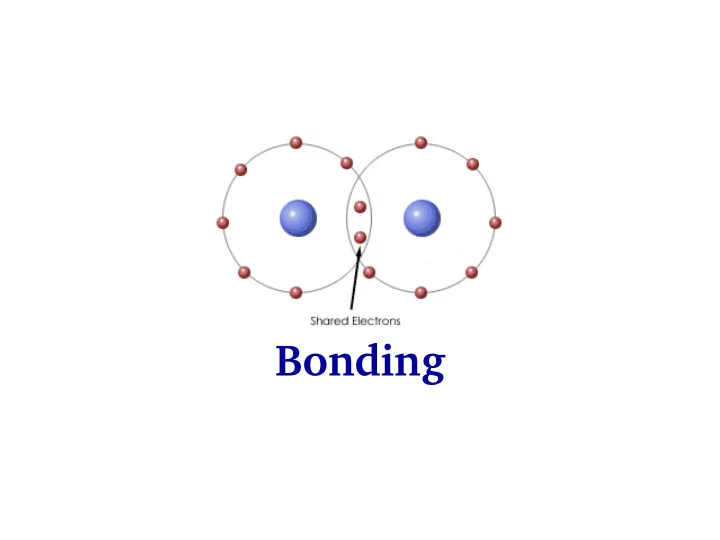
Bonding Bonding: H2+ and H2 molecules ( h h h ) 2 2 2 + = - - PowerPoint PPT Presentation
Bonding Bonding: H2+ and H2 molecules ( h h h ) 2 2 2 + = e- 2 2 2 - H H 2 A B e 2 m 2 m 2 m rA rB A B e r 2 2 2 e e e + + + Q Q Q R r r R HA HB A B ( h h
Bonding: H2+ and H2 molecules
+
- +
R HA HB rA rB r e-
+
- +
R HA HB r1A r1B r1 e-
- e-
r2B r2A r2
µ (
)
+ = −
∇ − ∇ − ∇ − − + h h h
2 2 2 2 2 2 2 2 2 2
2 2 2
A B e A B e A B
H H m m m e e e Q Q Q r r R
µ (
) = −
∇ − ∇ − ∇ − ∇ − − − + + h h h h
2 2 2 2 2 2 2 2 2 1 2 2 2 2 2 1 1 2 2 2 2 12
2 2 2 2
A B A B e e e e A B A B
H H m m m m e e e e Q Q Q Q r r r r e e Q Q r R
Born – Oppenheimer Approximation Nuclei are STATIONARY with respect to electrons ignore
µ (
)
+ = −
∇ − ∇ − ∇ − − + h h h
2 2 2 2 2 2 2 2 2 2
2 2 2
A B e A B e A B
e e e H H Q Q Q m m m r r R
µ (
)
+ = −
∇ − − + h
2 2 2 2 2 2
2
e e A B
e e e H H Q Q Q m r r R
µ (
)
+ = −
∇ − ∇ − ∇ − − + h h h
2 2 2 2 2 2 2 2 2 2
2 2 2
A B e A B e A B
e e e H H Q Q Q m m m r r R
Born – Oppenheimer Approximation ignore
µ (
) = −
∇ − ∇ − ∇ − ∇ − − − + + h h h h
2 2 2 2 2 2 2 2 2 1 2 2 2 2 2 2 2 1 1 2 2 12
2 2 2 2
A B e e A B e e A B A B
H H m m m m e e e e e e Q Q Q Q Q Q r r r r r R
µ (
) = −
∇ − ∇ − − − + + h h
2 2 2 2 2 2 2 2 2 2 2 1 2 1 1 2 2 12
2 2
e e e e A B A B
e e e e e e H H Q Q Q Q Q Q m m r r r r r R
Bonding: H2+ Molecule Difficult; but can be solved using elliptical polar co-ordinates
µ (
)
+ = −
∇ − − + h
2 2 2 2 2 2
2
e e A B
e e e H H Q Q Q m r r R
µ (
) ψ ψ
+ ×
= ×
2
( , ) ( ) ( , ) H H r R E R r R
Bonding: H2 molecule CANNOT be Solved For all the molecules except the simplest molecule H2+ the Schrodinger equation cannot be solved. We have approximate solutions
µ (
) ψ
ψ × = ×
2
( , ) ( ) ( , ) H H r R E R r R
µ (
) = −
∇ − ∇ − − − + + h h
2 2 2 2 2 2 2 2 2 2 2 1 2 1 1 2 2 12
2 2
e e e e A B A B
e e e e e e H H Q Q Q Q Q Q m m r r r r r R
Bonding For all the molecules except the simplest molecule H2+ the Schrodinger equation cannot be solved. We have only approximate solutions Valance-Bond Theory & Molecular Orbital Theory are two different models that solve the Schrodinger equation in different methods
Valance Bond Theory R=∞ R= Re Resonance H−−H ←→ H+−−H− ←→ H− −−H+ Inclusion of Ionic terms ψ ψ Ψ = ×
(1) (2) A B
ψ ψ ψ ψ Ψ = × + ×
(1) (2) (2) (1) A B A B
ψ
(1) A
ψ
(2) B
( ) ( )
ψ ψ ψ ψ λ ψ ψ λ ψ ψ λ λ
+ − − +
Ψ = × + × + × + × Ψ = Ψ + Ψ + Ψ
(1) (2) (2) (1) (1) (2) (1) (2) cov A B A B A A B B H H H H
Valance Bond Theory R= Re λ λ
+ − − +
Ψ = Ψ + Ψ + Ψ
cov H H H H
+
- +
R HA HB rA rB r e- A molecular orbital is analogous concept to atomic orbital but spreads throughout the molecule It’s a polycentric one-electron wavefunction (Orbital!) It can be produced by Linear Combination of Atomic Orbitals LCAO-MO Molecular Orbital Theory of H2+ ψ ψ − ∇ − − + = × ÷ h
2 2 2 2 2
2
e e A B
e e e Q Q Q E m r r R
+
- +
R HA HB rA rB r e- LCAO-MO Molecular Orbital Theory of H2+ ψ ψ − ∇ − − + = × ÷ h
2 2 2 2 2
2
e e A B
e e e Q Q Q E m r r R ψ φ φ = +
1 1 2 1
A B
MO s s
C C
( )
ψ φ φ φ φ = + +
2 2 2 2 2 1 1 2 1 1 2 1 1
2
A B A B
MO s s s s
C C C C = ⇒ = ±
2 2 1 2 1 2
Symmetry requirement C C C C
+
- +
R HA HB rA rB r e- + + +
- Molecular Orbital Theory of H2+
= ⇒ = ±
2 2 1 2 1 2
Symmetry requirement C C C C ( )
( )
ψ φ φ = = = + = +
1 2 1 1 1
1 1
A B
a a s s a A B
C C C C C s s ( )
( )
ψ φ φ = − = = − = −
1 2 2 1 1
1 1
A B
b b s s b A B
C C C C C s s
( )
ψ = + 1 1
Bonding a A B
C s s
( )
ψ
−
= − 1 1
Anti bonding b A B
C s s
Molecular Orbital Theory of H2+
( )
ψ = + 1 1
Bonding a A B
C s s
( )
ψ
−
= − 1 1
Anti bonding b A B
C s s
Bracket Notation
µ µ
φ φ τ φ φ δ φ φ τ φ φ
∗ ∗
= = = =
∫ ∫
i i
j i j ij allspace j i j ij allspace
d A d A A
= = = ≠ 1 (for ) 0 (for ) i j i j
Normalization 1 S is called Overlap-Integral ( ) ( )
[ ] [ ] [ ]
ψ ψ φ φ φ φ φ φ φ φ φ φ φ φ = = + + = + + + = + = + = −
2 1 1 1 1 1 1 2 1 1 1 1 1 1 1 1 2
1 1 1 2 2 1 2 2 Similarly 1 2 2
A B A B A A B B A B B A
a s s s s a s s s s s s s s a a b
C C C S C S C S φ φ φ φ φ φ φ φ = = = =
1 1 1 1 1 1 1 1
1
A A B B A B B A
s s s s s s s s
S
Overlap Integral Overlap-Integral S can be positive or negative or zero
Molecular Orbital Theory of H2+
[ ]
( )
[ ]
( ) ψ φ φ ψ φ φ = + + = − −
1 1 1 2 1 1
1 2 2 1 2 2
A B A B
s s s s
S S
µ µ
ψ ψ ψ ψ = =
1 1 1 1 2 2
E H E H
Molecular Orbital Theory of H2+
µ
[ ]
( ) µ
[ ]
( )
[ ] (
) µ ( )
[ ]
µ µ µ µ
ψ ψ φ φ φ φ φ φ φ φ φ φ φ φ φ φ φ φ = = + + + + = + + + = + + + +
1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1
1 1 2 2 2 2 1 2 2 1 2 2
A B A B A B A B A A B B A B B A
s s s s s s s s s s s s s s s s
E H E H S S E H S E H H H H S
Molecular Orbital Theory of H2+
µ
[ ]
( ) µ
[ ]
( )
[ ] (
) µ ( )
[ ]
µ µ µ µ
ψ ψ φ φ φ φ φ φ φ φ φ φ φ φ φ φ φ φ = = − − − + = − − − = + − − −
2 2 2 2 1 1 1 1 2 1 1 1 1 2 1 1 1 1 1 1 1 1
1 1 2 2 2 2 1 2 2 1 2 2
A B A B A B A B A A B B A B B A
s s s s s s s s s s s s s s s s
E H E H S S E H S E H H H H S
Molecular Orbital Theory of H2+
[ ]
( )
[ ]
( ) ψ φ φ ψ φ φ = + + = − −
1 1 1 2 1 1
1 2 2 1 2 2
A B A B
s s s s
S S
µ µ
ψ ψ ψ ψ = =
1 1 1 1 2 2
E H E H
[ ]
µ µ µ µ
φ φ φ φ φ φ φ φ = + − − −
2 1 1 1 1 1 1 1 1
1 2 2
A A B B A B B A
s s s s s s s s
E H H H H S
[ ]
µ µ µ µ
φ φ φ φ φ φ φ φ = + + + +
1 1 1 1 1 1 1 1 1
1 2 2
A A B B A B B A
s s s s s s s s
E H H H H S
Molecular Orbital Theory of H2+ Ĥ is Hermitian
[ ]
µ µ µ µ
φ φ φ φ φ φ φ φ = + + + +
1 1 1 1 1 1 1 1 1
1 2 2
A A B B A B B A
s s s s s s s s
E H H H H S
µ µ µ µ
φ φ φ φ φ φ φ φ φ φ φ φ = = = = = = = = =
1 1 1 1 1 1 1 1 1 1 1 1
i i j j i j j i i j j i
s s ii jj s s s s ij ji s s s s ij ji s s
H H H H H H H H S S
[ ]
µ µ µ µ
φ φ φ φ φ φ φ φ = + − − −
2 1 1 1 1 1 1 1 1
1 2 2
A A B B A B B A
s s s s s s s s
E H H H H S + + = = + + − − = = − −
1 2
2 2 2 2 1 2 2 2 2 1
ii ij ii ij ij ij ii ij ii ij ij ij
H H H H E S S H H H H E S S
Molecular Orbital Theory of H2+ + + = = + + − − = = − −
1 2
2 2 2 2 1 2 2 2 2 1
ii ij ii ij ij ij ii ij ii ij ij ij
H H H H E S S H H H H E S S
Molecular Orbital Theory of H2+
µ µ µ µ
= − ∇ − − + = − ∇ − − + ÷ = − + h h
2 2 2 2 2 2 2 2 2 2 2 2 1
2 2
e e A B e e A B e B
e e e H Q Q Q m r r R e e e H Q Q Q m r r R e e H H Q Q r R
µ µ
φ φ φ φ φ φ φ φ = = = + −
1 1 2 2 1 1 1 1 1 1 1
(or ) 1 1
i i i i i i i i
ii AA BB s s e s s s s s s B
H H H H H Qe Qe R r
Molecular Orbital Theory of H2+ Constant at Fixed Nuclear Distance J ⇒ Coulomb Integral
µ µ µ
φ φ φ φ φ φ φ φ φ φ φ φ φ φ = = = + − = + − = + − ×
1 1 2 2 1 1 1 1 1 1 1 2 2 1 1 1 1 1 1 1 2 2 1
(or ) 1 1 1
i i i i i i i i i i i i i i
ii AA BB s s e ii s s s s s s B e ii s s s s s s B ii s
H H H H H H Qe Qe R r Qe H H Qe R r Qe H E Qe J R φ φ φ φ = =
1 1 1 1
1 1
i i i i
s s s s B
J r
Molecular Orbital Theory of H2+ K ⇒ Exchange Integral Resonance Integral Constant K is purely a quantum mechanical
- concept. There is no classical
counterpart
µ µ µ
φ φ φ φ φ φ φ φ φ φ φ φ φ φ = = = + − = + − = + − ×
1 1 2 2 1 1 1 1 1 1 1 2 2 1 1 1 1 1 1 1 2 2 1
(or ) 1 1 1
i j i j i j i j i j i j i j
ij AB BA s s e ij s s s s s s B e ij s s s s s s B ij s
H H H H H H Qe Qe R r Qe H H Qe R r Qe H E S S Qe K R φ φ φ φ = =
1 1 1 1
1
i j i j
s s s s B
S K r
Molecular Orbital Theory of H2+
[ ] [ ] [ ] [ ] [ ] [ ] [ ] [ ] [ ] [ ]
+ = = + − + + − ÷ ÷ + + = + + + − + + + = + − + − = = + − − − − ÷ ÷ − − = − −
2 2 1 1 1 2 2 1 1 2 2 1 1 2 2 2 1 1 2 1
1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1
ii ij s s ij s s ii ij s s ij s
H H S E E Qe J E S Qe K S R R S Qe E E S S Qe J K S R Qe J K Qe E E R S H H S E E Qe J E S Qe K S R R S E E S S
[ ] [ ] [ ] [ ]
+ − − − − = + − −
2 2 2 2 2 1
1 1
s
Qe S Qe J K R Qe J K Qe E E R S
Molecular Orbital Theory of H2+
[ ]
( )
[ ] [ ] [ ]
( )
[ ] [ ]
ψ φ φ ψ φ φ = + + + = + − + = − − − = + − −
1 1 1 2 2 1 1 2 1 1 2 2 2 1
1 2 2 1 1 2 2 1
A B A B
s s s s s s
S Qe J K Qe E E R S S Qe J K Qe E E R S
Molecular Orbital Theory of H2+ Destabilization of Anti-bonding orbital is more than Stabilization of Bonding orbital J - Coulomb integral - interaction of electron in 1s orbital around A with a nucleus at B K - Exchange integral – exchange (resonance) of electron between the two nuclei.
[ ] [ ] [ ] [ ]
+ = + − + − = + − − ≤ ≤ < <
2 2 1 1 2 2 2 1
1 1 1 ; 0 &
s s
Qe J K Qe E E R S Qe J K Qe E E R S S J K
Molecular Orbital Theory of H2+
E1
1
E2