ASD SD Work
- rking Grou
- up – Cl
Clinical Tria rial De Design
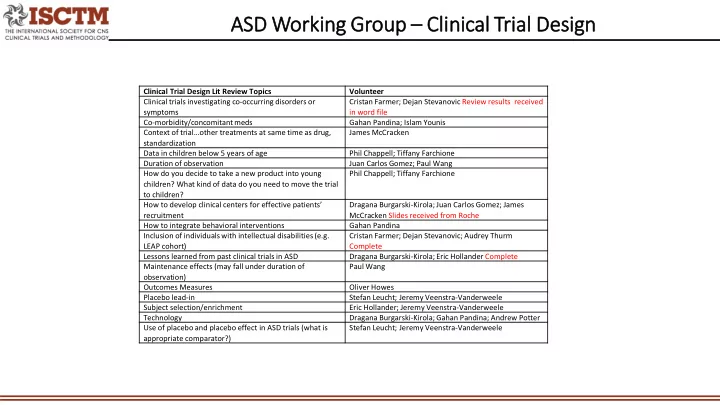
Clinical Trial Design Lit Review Topics Volunteer Clinical trials investigating co-occurring disorders or symptoms Cristan Farmer; Dejan Stevanovic Review results received in word file Co-morbidity/concomitant meds Gahan Pandina; Islam Younis Context of trial…other treatments at same time as drug, standardization James McCracken Data in children below 5 years of age Phil Chappell; Tiffany Farchione Duration of observation Juan Carlos Gomez; Paul Wang How do you decide to take a new product into young children? What kind of data do you need to move the trial to children? Phil Chappell; Tiffany Farchione How to develop clinical centers for effective patients’ recruitment Dragana Burgarski-Kirola; Juan Carlos Gomez; James McCracken Slides received from Roche How to integrate behavioral interventions Gahan Pandina Inclusion of individuals with intellectual disabilities (e.g. LEAP cohort) Cristan Farmer; Dejan Stevanovic; Audrey Thurm Complete Lessons learned from past clinical trials in ASD Dragana Burgarski-Kirola; Eric Hollander Complete Maintenance effects (may fall under duration of
- bservation)
Paul Wang Outcomes Measures Oliver Howes Placebo lead-in Stefan Leucht; Jeremy Veenstra-Vanderweele Subject selection/enrichment Eric Hollander; Jeremy Veenstra-Vanderweele Technology Dragana Burgarski-Kirola; Gahan Pandina; Andrew Potter Use of placebo and placebo effect in ASD trials (what is appropriate comparator?) Stefan Leucht; Jeremy Veenstra-Vanderweele