SLIDE 7 10/26/2017 7
Thank You!
- Acknowledgments
- Mark Blumenkranz, MD
- David Myung, MD, PhD
- Malvina Eydelman, MD
- Michelle E. Tarver, MD,
PhD
- Ron Schuchard, PhD
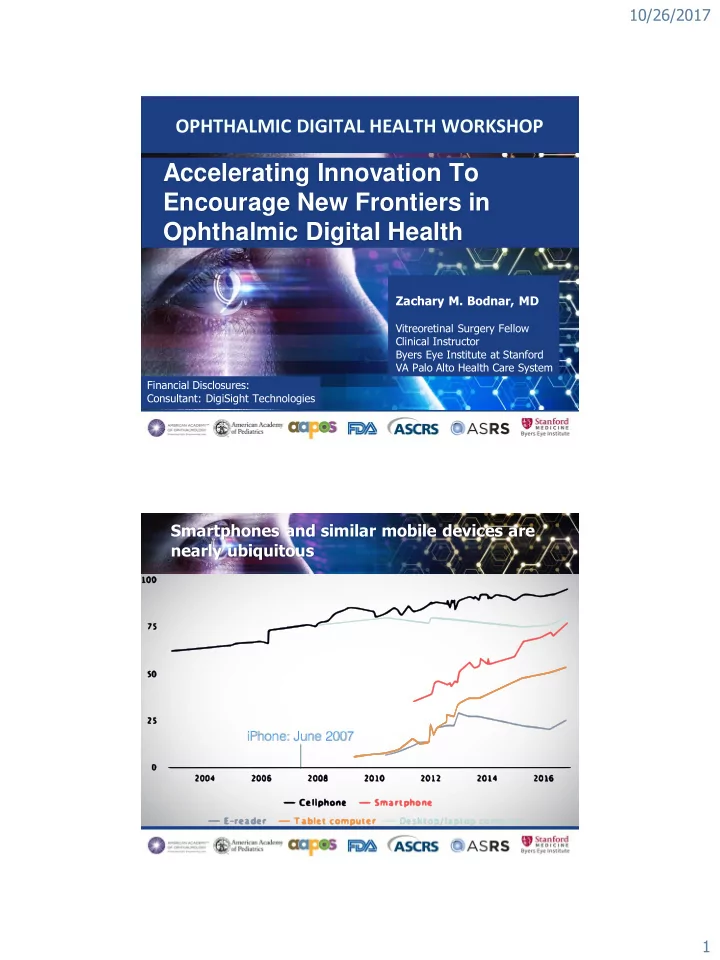
- Pew Research Center, Mobile Fact Sheet: http://www.pewinternet.org/fact-sheet/mobile/.
Accessed June 29, 2017
- What is a Medical Device?
https://www.fda.gov/AboutFDA/Transparency/Basics/ucm211822.htm. Accessed June 29, 2017.
- Federal Food Drug and Cosmetic Act:
https://www.fda.gov/regulatoryinformation/lawsenforcedbyfda/federalfooddrugandcosmeticac tfdcact/default.htm Accessed July 6, 2017.
- Digital Health: https://www.fda.gov/MedicalDevices/DigitalHealth/default.htm. Accessed June
29, 2017.
- Public Workshop - Mobile Medical Applications Draft Guidance, September 12-13, 2011:
https://www.federalregister.gov/documents/2011/08/12/2011-20574/mobile-medical- applications-draft-guidance-public-workshopAccessed July 6, 2017.
- Webinar - Final Guidance on “General Wellness: Policy for Low-Risk Devices” - September 1,
2016: http://www.fda.gov/MedicalDevices/NewsEvents/WorkshopsConferences/ucm515955.htm. Accessed June 29, 2017.
- 7. Requests for Feedback on Medical Device Submissions: The Pre-Submission Program and
Meetings with Food and Drug Administration Staff: http://www.fda.gov/downloads/medicaldevices/deviceregulationandguidance/guidancedocum ents/ucm311176.pdf. Accessed June 29, 2017.
Ref efer erences ences