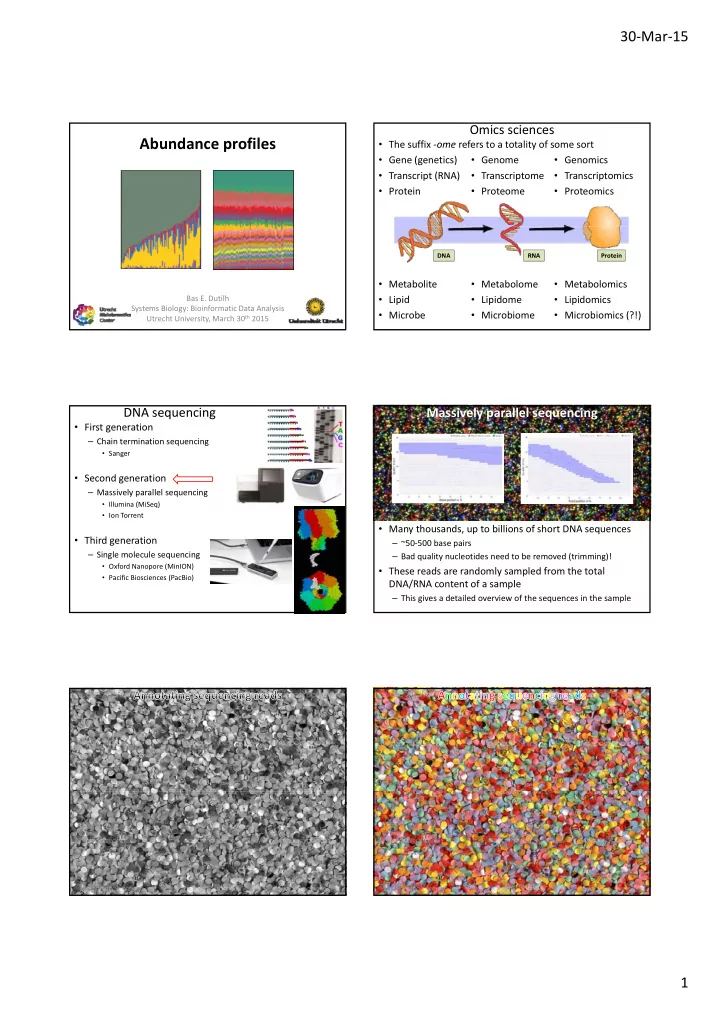
SLIDE 4 30‐Mar‐15 4
Microbial abundance in time
- Normalized and scaled microbial abundance values
– For example, presence of pathogens on rotting Arabidopsis leaves Mi b 2 Microbe 1
15 20 25
pression
0. 0.
Microbe 3 Microbe 2
5 10 15 1 2 3 4 5 6 7 8 9 10
Abundance/Exp Time/environments/samples… …or leaves…
0. 0. 0.0
Research setup
- 1. Design experimental conditions and sampling strategy
- 2. Extract DNA/RNA/protein
- 3. Sequence nucleotides or proteins
- 4. Quality control of sequencing reads or peptides
- 5. Annotate (e.g. align reads to database) and count
- 6. Normalize and scale the counts
- 7. Compare samples, clustering (next lecture)
- 8. Interpret results and perform verification experiments
Quantifying similarity between vectors
- Based on these measurements, which genes/microbes/etc are more
similar to each other?
15 20 25
pression
- Abundance/expression levels
are most similar between and
- Abundance/expression patterns
are most similar between and W di t
0. 0. 5 10 15 1 2 3 4 5 6 7 8 9 10
Abundance/Exp Time/Environments/Samples
- We can use a distance measure
to quantify the (dis‐)similarity between the lists – Many different distance measures exist
0. 0. 0.0
Distance matrices
x y x z y z
1 1 ‐ x 1 ‐ y 1 ‐ x 1 1 ‐ z 1 ‐ y 1 ‐ z 1
inverse inverse
distance = 1 ‐ similarity
Manhattan distance (levels)
0.265 0.265 0.799 0.799 0.534 0.534
d = |0.20 – 0.15| + dAB = |XA – XB| + |YA – YB|
1 0.20 0.15 0.12 2 0.17 0.15 0.09 3 0.16 0.16 0.08 4 0.20 0.15 0.11 5 0.20 0.16 0.12 6 0.17 0.16 0.10 7 0.16 0.15 0.08 8 0.20 0.15 0.12 9 0.18 0.16 0.11 10 0.16 0.15 0.08
|0.17 – 0.15| + |0.16 – 0.16| + |0.20 – 0.15| + |0.20 – 0.16| + |0.17 – 0.16| + |0.16 – 0.15| + |0.20 – 0.15| + |0.18 – 0.16| + |0.16 – 0.15| = 0.265 d = 0.799 d = 0.534
(YA – YB)2 dAB
2
= +
Euclidean distance (levels)
0.103 0.103 0.253 0.253 0.178 0.178
d 2 = (0.20 – 0.15)2 +
(YA – YB)2 dAB
2
dAB = (XA – XB)2 + (YA – YB)2
( A
B)
(XA – XB)2
(0.17 – 0.15)2 + (0.16 – 0.16)2 + (0.20 – 0.15)2 + (0.20 – 0.16)2 + (0.17 – 0.16)2 + (0.16 – 0.15)2 + (0.20 – 0.15)2 + (0.18 – 0.16)2 + (0.16 – 0.15)2 = 0.0105 d = 0.103 d = 0.253 d = 0.178
( A
B)
(XA – XB)2 1 0.20 0.15 0.12 2 0.17 0.15 0.09 3 0.16 0.16 0.08 4 0.20 0.15 0.11 5 0.20 0.16 0.12 6 0.17 0.16 0.10 7 0.16 0.15 0.08 8 0.20 0.15 0.12 9 0.18 0.16 0.11 10 0.16 0.15 0.08