SLIDE 1
1 Biologically-active Chiral Molecules HO HO OH HO O O O OH - - PDF document
1 Biologically-active Chiral Molecules HO HO OH HO O O O OH - - PDF document
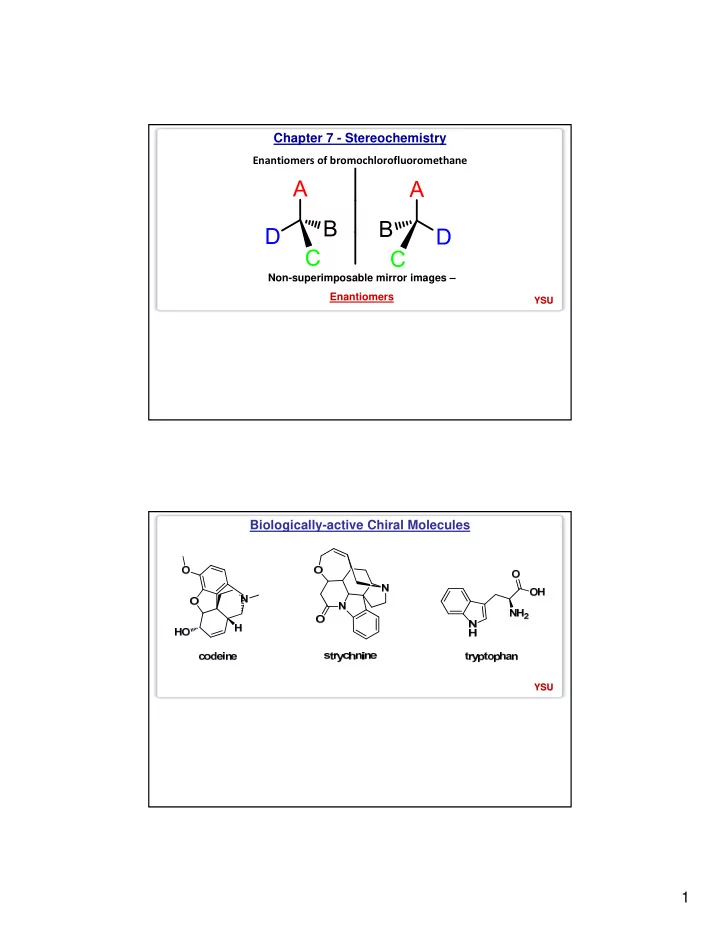
Chapter 7 - Stereochemistry Enantiomers of bromochlorofluoromethane Non-superimposable mirror images Enantiomers YSU YSU Biologically-active Chiral Molecules YSU YSU 1 Biologically-active Chiral Molecules HO HO OH HO O O O OH
SLIDE 2
SLIDE 3
3
YSU YSU
The Chirality Centre - Stereoisomerism
Carbon atom here is asymmetric C is a stereogenic center
YSU YSU
7.3 Symmetry in Achiral Structures
Mirror images of chlorodifluoromethane are superimposable Figure 7.2
Achiral i.e. not chiral
SLIDE 4
4
YSU YSU
Typical polarimeter setup : []D = 100 x (rotation)/(cell length) x (concentration)
7.4 Measurement of Optical Activity
YSU YSU
7.8 Enantiomers
- same physical properties except rotation of plane polarized light
- one enantiomer positive rotation (+) other negative rotation (‐)
SLIDE 5
5
YSU YSU
Which molecules contain chiral (stereogenic) centers?
The stereogenic C must have 4 different groups attached
YSU YSU
7.5 Absolute and Relative Configuration
Absolute Configuration – Actual arrangement of substituents in space (+)‐2‐butanol and (‐)‐2‐butanol, but which is which? Relative Configuration ‐ Configuration relative to another compound. Pre‐1951, compounds could be related to each other but the absolute configuration was not able to be determined.
SLIDE 6
6
YSU YSU
7.6 Nomenclature - Use of the Cahn-Ingold-Prelog System
S enantiomer R enantiomer R ‐ Rectus ‐ the clockwise arrangement of groups S ‐ Sinestre ‐ the counterclockwise arrangement of groups
YSU YSU
7.6 Nomenclature - Use of the Cahn-Ingold-Prelog System
SLIDE 7
7
YSU YSU
7.7 Fischer Projection Formulas
Figure 7.5
YSU YSU
7.9 Reactions that create a Chirality Center
Figure 7.6
SLIDE 8
8
YSU YSU
7.10 Chiral molecules with two Chirality Centers
Figure 7.7
YSU YSU
7.10 Representations of (2R, 3R)-dihydroxybutanoic acid
All the same molecule: (a) and (b) differ only by bond rotation (b) leads to correct Fischer projection
Conversion of “zig‐zag” picture to Fischer projection
Figure 7.8
SLIDE 9
9
YSU YSU
7.10 Chiral molecules with two Chirality Centers
Important stereochemical labels later, particularly in carbohydrate (sugar) chemistry and biochemistry
YSU YSU
7.10 Chiral molecules with two Chirality Centers
Applies to other cycles, including cyclohexane; increases the molecular diversity possible using simple structures
SLIDE 10
10
YSU YSU
7.11 Achiral molecules with two Chirality Centers
Figure 7.9
YSU YSU
Meso-2,3-butanediol
Figure 7.10
SLIDE 11
11
YSU YSU
7.11 Stereogenic Centers in Cholic Acid
Figure 7.11
YSU YSU
7.13 Reactions that produce diastereomers
Figure 7.12
SLIDE 12
12
YSU YSU
7.13 Reactions that produce diastereomers
Figure 7.12
YSU YSU
7.14 Resolution of a chiral substance into its enantiomers
Figure 7.13
SLIDE 13