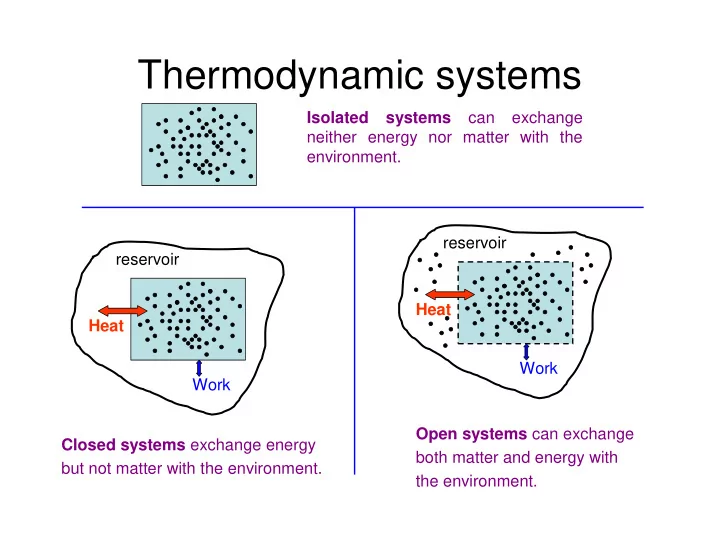
Isolated systems can exchange neither energy nor matter with the environment. Closed systems exchange energy but not matter with the environment. Heat Work reservoir Open systems can exchange both matter and energy with the environment. Heat Work reservoir
Thermodynamic systems Isolated systems can exchange neither energy - - PowerPoint PPT Presentation
Thermodynamic systems Isolated systems can exchange neither energy - - PowerPoint PPT Presentation
Thermodynamic systems Isolated systems can exchange neither energy nor matter with the environment. reservoir reservoir Heat Heat Work Work Open systems can exchange Closed systems exchange energy both matter and energy with but not
Quasi-static processes: near equilibrium Initial state, final state, intermediate state: p, V & T well defined Sufficiently slow processes = any intermediate state can considered as at thermal equilibrium. Thermal equilibrium means that It makes sense to define a temperature. Examples of quasi-static processes:
- isothermal:
T = constant
- isochoric:
V = constant
- isobaric:
P = constant
- adiabatic:
Q = 0
Quasi-static processes
Work in thermodynamics
Expansion: work on piston positive, work on gas negative Compression: work on piston negative, work on gas positive
Work during a volume change
∫
= ⇒ = = =
2 1
. .
V V
pdV W pdV Adx p dx F dW
Work in pV diagrams
Work done equals area under curve in pV diagram
Careful with the signs…
1st Law of Thermodynamics
W Q U − = ∆
Conservation of energy
Heat is positive when it enters the system Work is positive when it is done by the system Heat is negative when it leaves the system Work is negative when it is done on the system
1st Law of Thermodynamics
pdV dQ dU − =
Conservation of energy
Heat is positive when it enters the system Work is positive when it is done by the system Heat is negative when it leaves the system Work is negative when it is done on the system
) (
1 2 2
V V p W − =
- a. isochoric
- b. isobaric
- a. isobaric
- b. isochoric
) (
1 2 1
V V p W − =
∫
=
f i
V V pdV
W
isothermal
- The work done by a system depends on the initial and final states and
- n the path it is not a state function.
- Amount of heat transferred also depends on the initial, final, and intermediate
states it is not a state function either. (a) (b) (c)
State Functions
State Functions
1 2
U U W Q U − = − = ∆
The internal energy U is a state function: the energy gain (loss) only depends on the initial and final states, and not
- n the path.
Even though Q and W depend on the path, ∆U does not!
This pV–diagram shows two ways to take a system from state a (at lower left) to state c (at upper right):
- via state b (at upper left), or
- via state d (at lower right)
For which path is W > 0?
- A. path abc only
- B. path adc only
- C. both path abc and path adc
- D. neither path abc nor path adc
- E. The answer depends on what the system is made of.
CPS question
A system can be taken from state a to state b along any of the three paths shown in the pV– diagram. If state b has greater internal energy than state a, along which path is the absolute value |Q| of the heat transfer the greatest?
- A. path 1
- B. path 2
- C. path 3
- D. |Q| is the same for all three paths.
- E. not enough information given to decide
CPS question
Thermodynamic Processes:
- Adiabatic: no heat transfer (by insulation or by very fast process)
Q=0 → → → → U2 – U1 = -W
- Isochoric: constant volume process (no work done)
W=0 → → → → U2 – U1 = Q
- Isobaric: constant pressure process
p=const. →
→ → → W = p (V2 – V1)
- Isothermal: constant temperature process (heat may flow but very
slowly so that thermal equilibrium is not disturbed) ∆ ∆ ∆ ∆U=0,Q =-W only for ideal gas. Generally ∆
∆ ∆ ∆U,Q, W not zero
any energy entering as heat must leave as work
Thermodynamic processes
Isolated systems:
f i
U U U W Q = → = ∆ → = =
Cyclic processes P
i, f
V initial state = final state
W Q U = → = ∆
The internal energy
- f an isolated systems
remains constant
Adiabatic processes
W U Q − = ∆ → = 0
Expansion: U decreases Compression: U increases Energy exchange between “heat” and “work”
First Law for Several Types of Processes
More about cyclic processes
P
i, f
V
Work equals the area enclosed by the curves (careful with the sign!!!)
- A. Q > 0, W > 0, and ∆U = 0.
- B. Q > 0, W > 0, and ∆U > 0.
- C. Q = 0, W > 0, and ∆U < 0.
- D. Q = 0, W < 0, and ∆U > 0.
- E. Q > 0, W = 0, and ∆U > 0.
CPS question An ideal gas is taken around the cycle shown in this pV–diagram, from a to b to c and back to a. Process b → c is isothermal. For this complete cycle,
Important formulas
∆U=Q-W (1st law) (work during a volume change) pV=nRT (Ideal gas law) CV=f/2nR (Equipartition theorem)
∫
=
2 1
V V
pdV W
Isochoric process: V = constant
V P V1,2 1 2
2 2
nRT V p =
1 1
nRT V p =
2 1
= = →
∫
V V pdV
W
T C T T C Q
V V
∆ = − = ) (
1 2
Heat
reservoir During an isochoric process, heat enters (leaves) the system and increases (decreases) the internal energy.
(CV: heat capacity at constant volume)
T C Q U W Q U
V∆
= = ∆ → − = ∆
Ideal gas: isochoric process
Isobaric process: p = constant
V P V1 1 2
2 2
nRT pV =
1 1
T Nk pV
B
=
V p V V p pdV W
V V
∆ = − = = →
∫
) (
1 2
2 1
V2
T C T T C Q
p p
∆ = − = ) (
1 2
(CP: heat capacity at constant pressure)
V p T C W Q U
P
∆ − ∆ = − = ∆
→
During an isobaric expansion process, heat enters the system. Part of the heat is used by the system to do work on the environment; the rest of the heat is used to increase the internal energy. Heat Work reservoir
Ideal gas: isobaric process
Isothermal process: T = constant
V P V1 1 2
nRT pV =
1 2
ln
2 1 2 1 2 1
V V nRT V dV nRT dV V nRT pdV W
V V V V V V
= = = =
∫ ∫ ∫
V2
= ∆ ⇒ = ∆ U T
1 2
ln V V nRT W Q = = →
Expansion: heat enters the system all of the heat is used by the system to do work on the environment. Compression: the work done on the system increases its internal energy, all of the energy leaves the system at the same time as the heat is removed.
Ideal gas: isothermal process
Heat capacities of an Ideal gas
Consider an isobaric process p=constant
T C Q
p p
∆ =
From the 1st Law of Thermodynamics:
pdV dU dT C dQ
p p
+ = =
but
dT C dU
V
= pdV dT C dT C
V p
+ = ⇒ nRdT pdV Vdp pdV nRT pV = = + ⇒ =
From the Ideal gas law:
nR C C nRdT dT C dT C
V p V p
+ = ⇒ + = ⇒
Heat capacities of an Ideal gas
nR f C nR C C
V V p
2 = + = nR f Cp 2 2 + = ⇒ f f nR f nR f C C
V p
2 2 / 2 2 + = + = ⇒
f = #degrees
- f freedom
For a monoatomic gas f=3
67 . 1 3 / 5 ; 2 5 = = = ⇒
V p p
C C nR C
Molar heat capacities of various gases at (25 C)
1.05 8.76 3.29 27.36 36.12 H2S 1.02 8.51 3.41 28.39 36.90 N2O 1.02 8.45 3.39 28.17 36.62 CO2 Polyatomic 1.00 8.40 2.49 20.74 29.04 CO 1.01 8.39 2.52 20.98 29.37 O2 1.01 8.38 2.46 20.44 28.82 H2 1.00 8.32 2.50 20.80 29.12 N2 Diatomic 0.99 8.27 1.51 12.52 20.79 Xe 1.00 8.34 1.50 12.45 20.79 Kr 1.00 8.34 1.50 12.45 20.79 Ar 0.98 8.11 1.52 12.68 20.79 Ne 0.99 8.27 1.51 12.52 20.79 He Monoatomic (Cp-Cv)/R Cp-Cv Cv/R Cv Cp Gas
V P V1 1 2 V2
Adiabatic process: Q = 0
) , ( T V p p =
∫ ∫
= =
2 1 2 1
) , (
V V V V
dV T V p pdV W
T Nk pV
B
=
nRdT Vdp pdV nRT d pV d = + → = → ) ( ) (
pdV RdT f n pdV dT C pdV dW dU
V
− = → − = → − = − = 2
pdV f Vdp pdV 2 − = + f is the # of degrees of freedom
Ideal gas: adiabatic process
V P V1 1 2 V2
) , ( T V p p =
) 2 1 ( 2 = + + → − = + pdV f Vdp pdV f Vdp pdV
= + V dV p dp γ
constant ln ln ln
1 1 1 1 1 1
1 1
= = → = → = + → = + ∫
∫
γ γ γ γ
γ γ V p pV V p pV V V p p V dV p dp
V V p p
let , and dividing by pV ) 2 1 ( f + = γ
Ideal gas: adiabatic process (contd)
V P V1 1 2 V2
constant =
γ
pV
) ( 1 1 ) ( ) (
2 2 1 1 2 2 1 1 2 1
V p V p V p V p R C T T nC T nC U W
V V V
− − = − = − = ∆ − = ∆ − = γ
− − = →
− − 1 2 1 1 1 1
1 1 ) 1 ( 1
γ γ γ
γ V V V p W
constant
1 1
= =
γ γ
V p pV
Ideal gas: adiabatic process (contd)
Using:
V P V1 1 2 V2
constant =
γ
pV
γ γ 2 2 1 1
V p V p =
2 2 2 1 1 1
nRT V p nRT V p = =
2 1 2 1 2 1
T T V V p p = →
γ γ 1 2 2 1
V V p p = →
2 1 1 1 1 2
T T V V = →
− − γ γ
- r
constant
1 1
= =
γ γ
V p pV
constant
1 2 2 1 1 1
= =
− − γ γ
V T V T
Ideal gas: adiabatic process (contd)
V P V1 1 2 V2
constant =
γ
pV
constant
1 1
= =
γ γ
V p pV
constant
1 2 2 1 1 1
= =
− − γ γ
V T V T
nRT pV =
During an adiabatic expansion process, the reduction of the internal energy is used by the system to do work on the environment. During an adiabatic compression process, the environment does work on the system and increases the internal energy.
Ideal gas: adiabatic process (contd)
− − =
− − 1 2 1 1 1 1
1 1 ) 1 (
γ γ γ
γ V V V p W
When an ideal gas is allowed to expand isothermally from volume V1 to a larger volume V2, the gas does an amount of work equal to W12. If the same ideal gas is allowed to expand adiabatically from volume V1 to a larger volume V2, the gas does an amount of work that is
- A. equal to W12.
- B. less than W12.
- C. greater than W12.
- D. either A., B., or C., depending on the ratio of V2 to V1.
CPS question
Quasi-static process Character
U ∆
W
Q
adiabatic
= Q
W U − = ∆
isothermal T = constant
= ∆U
isochoric isobaric V = constant p = constant
Q U = ∆
T C Q
V∆
=
= W
V p W ∆ =
W Q U − = ∆
T C Q
P∆
=
1 2
ln V V nRT W =
W Q =
) 1 1 ( ) 1 ( 1
1 1 1 2 1 1 − − −
− =
γ γ γ
γ V V V p W
= Q