The Laboratory of Atmospheric Chemistry The Laboratory of - - PowerPoint PPT Presentation
The Laboratory of Atmospheric Chemistry The Laboratory of - - PowerPoint PPT Presentation
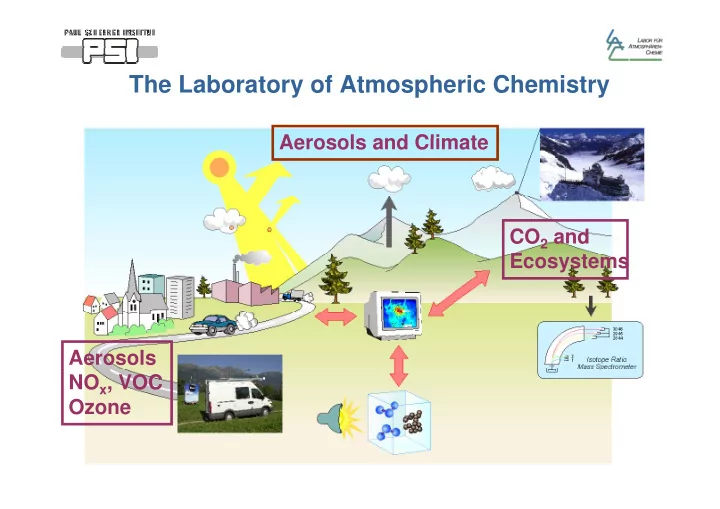
The Laboratory of Atmospheric Chemistry The Laboratory of Atmospheric Chemistry Aerosols and Climate Aerosols and Climate CO 2 and E Ecosystems t Aerosols NO x , VOC Ozone Aerosols Definition: PM10 = Particles with aerodynamic diameter
Aerosols
Examples:
Ammonium sulfate: ca 0 1 µm
Definition: PM10 = Particles with aerodynamic diameter <10µm Examples:
Diesel soot: ca. 0.1 µm Ammonium sulfate: ca. 0.1 µm Pollen: 10 - 100 µm Sea salt: 0.2 - 10 µm Mineral dust: 0.2 - 10 µm
How big are aerosol particles?
Size relationships
x 20’000 x 20’000 Diesel soot 100 Nanometer (nm) Pin head
2’000’000 nanometer (nm) =
Hot air balloon ( )
= 0.1 micrometer (µm) = 0.0001 millimeter (mm) = 2’000 micrometer (µm) =
2 Millimeter (mm)
40’000’000 micrometer (µm) = 40’000 millimeter (mm) =
40 Meter (m) 40 Meter (m)
In one cubiccentimeter of air: typically 10‘000 particles
Aerosols Aerosols Aerosols Aerosols
- Primary and secondary particles and size distributions
- Instrumentation
- Climate
- Health
- Source identification
Primary particle emissions in 2000 (Tg yr-1)
IPCC, 2001
Formation of secondary aerosol
- Sulfate, homogeneous reaction:
SO2 + •OH + M → HO•SO2 + M HO•SO2 + O2 + M → HOO• + SO3 + M SO3 + H2O → H2SO4
- Sulfate heterogeneous reaction:
- Sulfate, heterogeneous reaction:
SO2(g) ↔ SO2(aq) SO2(aq) + H2O ↔ HSO3
- (aq) + H3O+(aq)
Oxidation with H2O2, Ozon, NO2, …
- Nitrate, homogeneous reaction :
NO2 + •OH → HNO3 NO2 + OH → HNO3 NH3 (g) + HNO3 (g) ↔ NH4NO3 (s)
- Nitrate, heterogeneous reaction :
NO O NO ( ) O NO2 + O3 → NO3 (g) + O2 NO3 (g) + NO2 + M → N2O5 (g) + M N2O5 (g) + H2O (aq) → 2 HNO3 (aq)
- Organics:
VOC + OH, O3, … SOA (secondary organic aerosol)
Source strengths of sulfate and
- rganic carbon (kg m-2 hr-1)
IPCC, 2001
Source strengths of black carbon, mineral dust and sea salt (kg m-2 hr-1)
IPCC, 2001
Size distributions of aerosol particles
Aerosol Size Distribution
40
x103
Nucleation Mode
20 30
Number gDp), cm-3x
Mode
10
N (dN/dlog
Aitken Mode
40
m3/cm3
Droplet Submode Acc m lation
20 30
Volume dlogDp), µm
Condensation Submode Submode Coarse Mode Accumulation Mode
10
(dV/d
0.01 0.1 1 10
Diameter (micrometers)
Traffic emissions: Influence of dilution temperature
1.0E+10
Tair=15C
1.0E+09
m
3
) Tair=25C Tair=35C Tair=42.5C
1 0E+07 1.0E+08
- gDp (#/cm
1 0E+06 1.0E+07
DN/ Dl
1.0E+06 1 10 100 1000
Dp ( nm)
Kittelson et al. (2000)
Size distribution in and around Zürich (day, night): ( y, g )
smallest particles are 10 times more abundant in the city compared to the country side and 100 smallest particles are 10 times more abundant in the city compared to the country side and 100 times more abundant duirng the day than in the night times more abundant duirng the day than in the night
10
6 4
10
5
10
3
10
4
(cm
- 3)
10
1
10
2
N/d(logD)
10 10
1
Urban Area: (Downtown Zьrich) Day (SMPS)
dN
Rural Region: (Zьrcher Oberland) Day (SMPS)
10
- 2
10
- 1
Night (SMPS) Day (OPC) Night (OPC) Night (SMPS) Day (OPC) Night (OPC) 10 100 1000 10000
10
Dp (nm)
10 100 1000 10000
Bukowiecki et al., 2002
Nucleation and Growth in Pittsburgh (August 11 2001) (August 11, 2001)
- S. Pandis
Size distributions in Milan: Evidence for primary and secondary particle formation
Time
Baltensperger et al., 2002
Secondary organic aerosol production Secondary organic aerosol production
Terminology: Terminology: Primary versus secondary
- Primary particles: directly emitted to the atmosphere
- Secondary particles: formed in the atmosphere by condensation
- Secondary particles: formed in the atmosphere by condensation
(nucleation and growth) after chemical transformation
- How about oxidized primary particles ?
aged primary (not secondary as Fuzzi et al. 2006 suggested)
- How about primary particles that evaporate on dilution and condense
after oxidation (Robinson et al., 2007) ? secondary
Measurement Techniques Measurement Techniques
- Number
- Number size distribution
- Mass
- Optical properties
- Aerosol composition (off-line / on-line)
Condensation Particle Counter (CPC) ( )
Example: TSI CPC 3010 Lowest detectable diameter: D = 10 nm Lowest detectable diameter: D = 10 nm Maximum particle concentration: 104 cm-3
Measuring the electrical mobility with a Differential Mobility Analyzers (DMA)
Measurable sizes: D=3-150 nm or 20-900 nm A scan is possible within 60-300 seconds A scan is possible within 60 300 seconds Principle:
- Defined electrical charging of the particles with radiocative source
Defined electrical charging of the particles with radiocative source
- The aerosol flows laminarily through cylindric condensator
- An electrical field force the particles depending on their electrical mobility to
An electrical field force the particles depending on their electrical mobility to move toward the inner electrode.
- Particles of a specific mobility (b=vp/E) are sucked through a gap at the
inner electrode and are detected (typically by a CPC) afterwards.
F t A l Fast Analyzers:
- EEPS
Engine Exhaust Particle
- Spectrometer. Scan time typ. 2 sec.
- FPS
Fast Particle Spectrometer
- FPS
Fast Particle Spectrometer (similar to EEPS)
- FMPS
Fast Mobility Particle Sizer (ambient air version of EEPS) Engine Exhaust Particle Sizer Spectrometer (EEPS, TSI) Fast Particle Spectrometer (DMS500, Cambustion)
Aerodynamic particle sizer
Mobile laboratory
CPC
The PSI mobile laboratory
CPC FMPS MAAP AMS CO2 CO2
Aerosol size distribution (5.6-560 nm) in an Alpine valley
10
6
5x10
6
4
# cm-3 Highway Village
80x10
9
60
m-3 cm-3 Highway Village
3 2
dN/dlogdp / #
40
dV/dlogdp/ nm
1
6 7 8 9
10
2 3 4 5 6 7 8 9
100
2 3
d /
20
6 7 8 9
10
2 3 4 5 6 7 8 9
100
2 3
Alpine Valley
dp / nm dp / nm
Alpine Valley Riviera 2005 Winter
- Consistent picture : Nanoparticle concentrations <30
Consistent picture : Nanoparticle concentrations 30 nanometers very high on highway
- In villages : much lower nanoparticle concentrations, in case of
high wood burning contribution higher volume concentration high wood burning contribution, higher volume concentration
Particulate mass Particulate mass
- Filters and gravimetric analysis
O li B t t TEOM
- On-line : e.g. Betameters, TEOM
Tapered element oscillating microbalance (TEOM)
Optical instruments e.g. light absorption by aethalomete
I0
⎞ ⎜ ⎛ ⎞ ⎜ ⎛ 0 I I x b
abs
e I I
⋅ −
=
“Lambert-Beer’s Law”
Aerosol particles
⎟ ⎠ ⎞ ⎜ ⎜ ⎝ ⎛ × = ⎠ ⎞ ⎜ ⎝ ⎛ × =
s r
I I ln 100 I I ln 100 ATN
“Optical attenuation”
⎟ ⎟ ⎠ ⎞ ⎜ ⎜ ⎝ ⎛ − ⎟ ⎟ ⎠ ⎞ ⎜ ⎜ ⎝ ⎛ = ∆ ) ( ) ( ln ) ( ) ( ln
1 1 2 2
t I t I t I t I ATN
s r s r
Quartz filter
x I S R For the time interval ∆t=t2-t1 :
⎠ ⎝ ⎠ ⎝ ) ( ) (
1 2 s s I S R The attenuation coefficient (filtered aerosol) The absorption coefficient (airborne aerosol)
t ATN Q A bATN ∆ ∆ =
babs
Correct ion needed, e. g.
The attenuation coefficient (filtered aerosol) The absorption coefficient (airborne aerosol)
- Weingart ner et al. , 2003
- Arnot t et al. , 2005
The absorption exponent α
Absorption exponent α: a measure
babs ∝ λ−α
10.0 12.0
m)
- f the spectral variation in
aerosol light absorption
6 0 8.0
babs (950nm
Example power law fit (λ-2.0) Wood burninga λ-1.8 to -2.2
4.0 6.0
babs(λ) /
g Traffic, diesel soota,b λ-1.0 to -1.1
0.0 2.0 350 450 550 650 750 850 950
a Kirchstetter et al. 2004
b Schnaiter et al. 2003 & 2005 350 450 550 650 750 850 950
λ [nm]
Enhanced UV- absorpt ion due t o t he presence of wood smoke Enhanced UV absorpt on due t o t he presence of wood smoke
Aerosol Chemical Analysis
Offline: Part 1 Offline: Part 2 Online / Direct Sampling Interface Extraction
Chemical Analyzer Chemical Analyzer Analyzer
Data Data
Pump
SO4 = 7.2 µg m-3
2-
Data
SO4
2-
Data Time
Slide courtesy of Jose-Luis Jimenez
Examples of off-line analysis
- Ion chromatography (NH4, NO3, SO4, organic acids)
- X-ray fluorescence, Particle Induced x-ray emission
(PIXE) Inter Coupled Plasma mass spectrometry (ICP (PIXE), Inter Coupled Plasma mass spectrometry (ICP- MS), Neutron activation, Atomic Asorption Spectroscopy (AAS) (elemental analysis: K, S, Pb, Zn, ..) (AAS) (elemental analysis: K, S, Pb, Zn, ..)
- GC- / LC-MS (organic compounds: e g marker
GC / LC MS (organic compounds: e.g. marker compounds hopanes, levoglucosan,)
- IR / UV / proton-NMR- spectroscopy : functional groups
- Mass spectrometry in general : isotope analysis,
- ligomers and more
On Line Analysis On-Line Analysis
- Semi-online: EC-OC (separation of black/elemental
b f i b ) carbon from organic carbon)
Miyazaki et al. , JGR, 2007
On line analysis On-line analysis
- ATOF-MS
- Aerodyne Aerosol mass spectrometer
- ..
Example of Aerosol mass spectrometer measurements together with some black Example of Aerosol mass spectrometer measurements together with some black carbon measurements by an Aethalometer carbon measurements by an Aethalometer
70 60 m
- 3)
Reiden
30 25 m
- 3)
Zurich
50 40 30 20 ss Concentration (µg
Organic Nitrate Ammonium Sulfate BC
20 15 10 ss C oncentration (µg
Organics Nitrate Sulphate Ammonium BC
10 M as 29.01.2006 31.01.2006 02.02.2006 04.02.2006 06.02.2006 08.02.2006 10.02.2006 12.02.2006 Date & Time 100 % 5 M as 08.01.2006 11.01.2006 14.01.2006 17.01.2006 20.01.2006 23.01.2006 Date & Time 100 % 80 60 40 ractional Contribtion 80 60 40 actional Contribution 20 Fr 29.01.2006 31.01.2006 02.02.2006 04.02.2006 06.02.2006 08.02.2006 10.02.2006 12.02.2006 Date & Time 20 Fra 08.01.2006 11.01.2006 14.01.2006 17.01.2006 20.01.2006 23.01.2006 Date & Time
Ti l ti i t d t 6 d t l d t ti li it Time resolution: minutes down to 6 seconds at low detection limits
Aerodyne aerosol mass spectrometer output: size distribution
15 10 Dva (µg m
- 3)
Organics Nitrate Sulphate Ammonium
5 dM/dlogD
4 5 6 7 8 9
100
2 3 4 5 6 7 8 9
1000
2
- Aerosol mass size
distributions
100 1000 dM/dlogDva (µg m
- 3)
Time Resolution Example - Fi Pl f i ft Fire Plumes from aircraft
5000 400 100 4000 400 300 PM1 80 m
- 3)
Altitude AMS total Mass PM1 scattering 3000 ude (ft) 300 Light Scatte 60 Loading (µg rf09_fire2 2000 Altitu 200 ring (Mm
- 1)
40 rosol Mass L rf09_fire1 rf09_fire3 1000 100 20 Ae 8:08 PM 3/23/06 8:10 PM 8:12 PM 8:14 PM 8:16 PM 8:18 PM 8:20 PM UTC UTC
3 Fires in the Yucatan Peninsula - MILAGRO field campaign
Organic Aerosol Analysis in Perspective
HR-AMS
100 alyzed)
Perfect Instrument EC/OC AMS
PBTDMS PILS-OC
HR-AMS Perfect Instrument EC/OC AMS
PBTDMS PILS OC
ion
High
Organic Aerosol Analysis in Perspective 80 60 Mass Ana
PILS-OC FTIR NMR VUV PILS-OC FTIR NMR VUV
Resoluti
40
s (% of M
CI, EA
2D-GCMS
CI, EA
2D-GCMS
and Size
20
eteness
Useless Instrument GC/MS
Tradeoff
Useless Instrument GC/MS
Time a
Low Comple
Class lasses lasses pecies ular ID
Instrument
Tradeoff
Instrument
Correlation btw what is C
One Few C Many C ses to Sp Molecu
Correlation btw what is NOT detected and sources led to missing
Clas
Selectivity SOA for decades
Slide courtesy of Jose-Luis Jimenez
Direct and indirect aerosol effect on climate Direct and indirect aerosol effect on climate
Direct effect: Scattering and absorption of incoming sunlight by aerosol particles Indirect effect: The number concentration of cloud condensation nuclei (CCN) influences the cloud droplet size d th b h th l d and thereby changes the cloud albedo and lifetime
Indirect aerosol effect
Indirect effect
Number of CCN i fl th d l t Large droplets Weak reflection
Small droplets Strong reflection
influences the droplet number and size (Twomey-Effect) and thereby the cloud thereby the cloud albedo and lifetime.
‘Ship tracks’
visualise the indirect effect visualise the indirect effect
Satellitenaufnahme ( Wellenlänge: 3.7 µm)
Indirect aerosol effects
IPCC (2007)
Climate forcing 2000 relative to 1750
IPCC (2001), www.ipcc.ch
Radiative forcing of climate between 1750 and 2005 Radiative forcing of climate between 1750 and 2005
No CO2 time scale is given,as its removal from the atmosphere involves a range of processes that can span long time scales :
IPCC (2007)
Radiative forcing of climate between 1750 and 2005 for different aerosol components 1750 and 2005 for different aerosol components
IPCC (2007)
Aerosol direct radiative forcing – Comparison of different models p
IPCC (2007)
Radiative forcing due to the cloud albedo effect – Comparison of different models cloud albedo effect Comparison of different models
IPCC (2007)
Total Aerosol Optical Depth (MODIS satellite) modelling
IPCC (2007)
Satellite retrieved AOD over Europe: Aerosols show high spatial variability Aerosols show high spatial variability
Polluted Polluted Clean
Robles Gonzales et al., GRL 27, 955 (2000)
Pathways of the Traditional Warm Indirect Aerosol Effect d h Gl i i I di A l Eff
+
and the Glaciation Indirect Aerosol Effect
Cloud albedo + + _ Cloud cover and lifetime + _ Precipitation _ + + + Cloud droplets + + +
Mixed phase cloud hydrometeors
+ + Ice crystals + Aerosol particles Cloud cond. nuclei + + Ice nuclei + Human activity
Lohmann, GRL, 2002
Evolution of particles in cloud: Bergeron-Findeisen process
Cloud Aerosol particle Cloud Ice crystals droplet Snow S o Aerosol particle Rain
Saturation Vapor Pressure (SVP) difference: SVP (ice) < SVP (liquid) ⇒ Flux of water vapor from liquid droplets to ice crystals The WBF mechanism converts many small supercooled drops to only few, large ice crystals, thus changing cloud radiative properties and enhancing precipitation.
Aerosols and Health Aerosols and Health
Aerosols is not a recent problem: The lung of ‘Ötzi‘ black: soot red: soot
Source: www.ecocouncil.dk
Air pollution and mortality during London winter smog in 1952 during London winter smog in 1952.
Adapted from Wilkins (1954)
Increased mortality correlates best with PM2.5 (fine particles)
Dockery et al. (1993)
Epidemiology: clear relationship between PM2.5 (or PM10) and mortality between PM2.5 (or PM10) and mortality
Bold: 1974-1989 (Dockery et al., 1993) ( ) Italic: 1990-1998 (Laden et al., 2006) Laden et al., 2006 CH: 3700 premature deaths per year
Different health effects of PM10
- Well established: Of all air pollutants, PM10 (and even more so PM2.5) show
best correlation with increased mortality best correlation with increased mortality
- Increase of mortality with increase of PM10 by 10 µg/m3 (Laden et al., 2000):
- Traffic:
3.4%
- Coal combustion:
1 1% Coal combustion: 1.1%
- Mineral dust: ~0%
Influence of
- Chemical composition (metals radicals organic compounds acidity)
- Chemical composition (metals, radicals, organic compounds, acidity)
- Biological constituents (allergens, endotoxin)
- Morphology (effect of asbestos)
- Size distribution: smaller particles have greater surface area per unit mass
Size distribution: smaller particles have greater surface area per unit mass
- Number concentration
- …
- Mechanism not well known yet
- Mechanism not well known yet
Loss in life expectancy
attributable to anthropogenic PM2 5 [months] attributable to anthropogenic PM2.5 [months] 2000 2020 2020 C t l i l ti MTFR Current legislation MTFR
Loss in average statistical life expectancy due to identified anthropogenic PM2.5 Calculations for 1997 meteorology Provisional estimates with generic assumption on urban increment of PM
- M. Amann, IIASA
Cytotoxicity of wood smoke from bad combustion: N Klippel Verenum bad combustion: N. Klippel, Verenum
Average composition in Zürich in summer and winter
13% 7% 7%
11% 1% 14%
13%
32% 27% 27%
Black Carbon O i
60%
Zürich (July)
15%
Zürich (January) Organic mass Nitrate Sulfate Ammonium Ammonium
Concentration of solid ammonium nitrate f ti f t t as a function of temperature
7 µg/m3 NH3 µg
3
26.5 µg/m3 HNO3 RH = 30% Gas phase conc.
Chemical composition in Zürich summer and Positive Matrix Factorization of the organic matter
Organic mass O ga c ass
13% 7% 7% 13%
Black Carbon O i
60%
Zürich (July) Organic mass Nitrate Sulfate Ammonium
OOA: Secondary organic aerosol
Lanz et al., ACP (2007) Ammonium
HOA: mostly traffic
8 1.6 wood burning (a=0.6) [µg m
- 3]
Plausibility of solution including 3 factors
29
Roveredo MS: R2 = 0.93 wood burning (modelled) vs CO (measured): R2 = 0.78
6 4 2 1.6 1.4 1.2 1.0 0.8 0.6 wood burning (a=0.6) [µg m
- 3]
CO [ppm] 0.4 0.3 0.2 0.1
- norm. intensity
wood burning aerosol 43 55 60 18 27
chestnut MS: R2 = 0.87
2 09.01.2006 13.01.2006 17.01.2006 21.01.2006 25.01.2006 dat 0.6 0.4 0.2 8 4 OOA (a=0.6) [µg m
- 3]
0.6 44 18 0.1 0.0 140 130 120 110 100 90 80 70 60 50 40 30 20 10 m/z 60 73
G
OOA (modelled) vs. AMS-ammonium (measured) R2 = 0.72
6 4 2 3 2 1 AMS-NH4 [µg m
- 3]
0.6 0.5 0.4 0.3 0.2 0.1
- norm. intensity
- xygenated organic aerosol (OOA, type I)
55 29
culated G culated F
aged rural MS: R2 = 0.93 fulvic acid MS: R2 = 0.87
09.01.2006 13.01.2006 17.01.2006 21.01.2006 25.01.2006 dat
6 5 300 250 HOA (a=0.6) 0.14 0.12 hydrocarbon-like organic aerosol (HOA) 57 55 43 41 0.0 140 130 120 110 100 90 80 70 60 50 40 30 20 10 m/z
cal calc
4 3 2 1 200 150 100 50 NOx [ppb] 0.10 0.08 0.06 0.04 0.02 0.00
- norm. intensity
67 69 71 27 29 83 81 85 91 95
HOA (modelled) vs. NOx (measured): R2 = 0.70
09.01.2006 13.01.2006 17.01.2006 21.01.2006 25.01.2006 dat 0.00 140 130 120 110 100 90 80 70 60 50 40 30 20 10 m/z
Lanz et al., ES&T, accepted
C b ti t i
14C
l i Carbon apportionment using 14C analysis Estimation of fossil and non-fossil SOA contribution
Z_meanPM10
Zürich (mean)
Use of AMS analysis :
38%
Wood burning
ECbb 4% ECfossil
Zürich (mean)
y
- wood burning 38%
- HOA 7%
(biomass burning) 38%
Traffic Secondary (fossil) S d
OM
17%
Assumptions :
- only SOA, HOA and wood
7% 17%
Secondary (non-fossil)
OCfossil 23% OCnonfossil 56%
burning present
- OM/OC=2 for wood
burning and SOA and
19%
EC (fossil)
burning and SOA and OM/OC=1.2 for HOA
EC (fossil) EC (non-fossil)
EC
81%
Lanz et al., accepted in ES&T
Smogchamber results of Carnegie Mellon Smogchamber results of Carnegie Mellon
S h b lt
- Smog chamber results
indicate that wood burning emissions is doubled after
- nly 2 hours of chemistry
Roveredo in an Alpine valley, Januar 2005
ECfossil ECbiomass 9%
06:00 – 14:00
13% OCfossil OCfossil 12% OCnonfossil 66%
ECfossil ECbiomass 11%
Roveredo (GR)
8%
18:00 – 02:00
OCnonfossil 81%
Szidat et al., GRL, 2007
A di l l f th b t i l OC/EC Average diurnal cycle of the carbonaceous material, OC/EC and wood burning versus traffic contributions
Sandradewi et al., ES&T, 2008
Aerosol mass spectra
12 8 4 Levoglucosan 29 44 60 73 167 137
Levoglucosan
12 8 Chestnut, poor burning 29 43 60 167 137
Wood burner (emissions) chestnut, very inefficient burning
4 anics 12 8 Night Filter, March 29 43 73 115 137 167
Night period in Roveredo in March, more than 80% of OC non fossil
% to total orga 4 12 60 73 115 137 167 Averaged nights, Dec 29 43
more than 80% of OC non-fossil Average in Roveredo over the
8 4 12 43 60 73 115 137 167
g whole December
12 8 4 29 44 55 69 81 91 Haerkingen, May
Mass spectra from a Motorway site in May
200 160 120 80 40 m/z
Alfarra et al. ES&T (2007)
SOA formation from TMB (at 50% RH) SOA formation from TMB (at 50% RH) Trimethylbenzene + NOx + light →→ Secondary Organic Aerosol
Observation of SOA oligomerization by Laser Desorption Ionization
1500 1000
mV)
500
Intensity (m
B)
Time after lights on: 3.5 h
1500 1000
mV)
500
Intensity (m
C)
4.5 h
4000 3000
V)
3000 2000 1000
Intensity (mV
D)
6.5 h
800 700 600 500 400
mass m/z
Kalberer et al., Science (2004)
Comparison of smogchamber aerosols with ambient sample
. a.u
trimethylbenzene-SOA
800 700 600 500 400 300
Pattern of Zurich points to α pinene
a.u.
α− pinene-SOA
points to α-pinene rather than TMB (or other anthrop. )
800 700 600 500 400 300
precursors)
a.u.
Downtown Zurich
Baltensperger et al.
800 700 600 500 400 300
p g Faraday Disc. (2005)
Worldwide AMS measurements
- f the chemical composition
Zhang et al., GRL 2007
Models underestimate SOA
Volkamer et al. GRL 2006
SUMMARY SUMMARY
A significant progress was made in last years but many challenges remain: B tt i t t ti ( h ll l
- Better instrumentation (cheaper, smaller, more long-
term, more specific, more precise, … ) is still needed
- Long term chemical composition of aerosols is needed
- Long-term chemical composition of aerosols is needed
- Secondary organic aerosol formation needs to be
understood and implemented in models understood and implemented in models
- Health effects : more specific to size and chemical
composition composition
- Climate : Indirect effects are not quantitatively