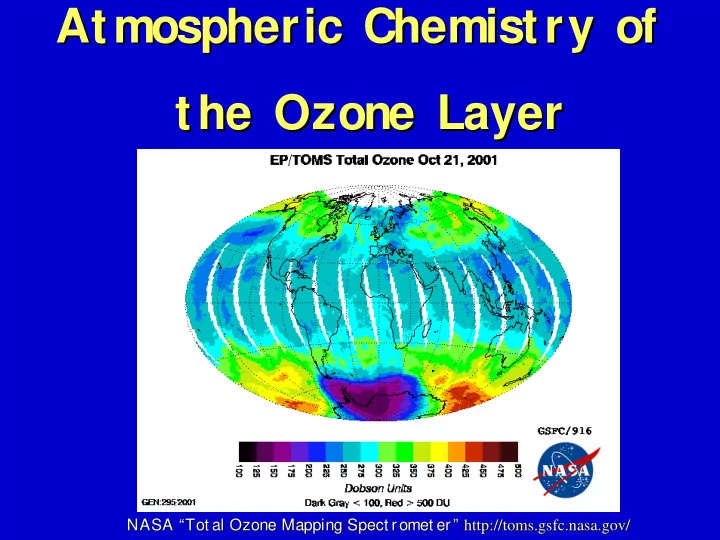
SLIDE 1 Atmospheric Chemistry of Atmospheric Chemistry of the Ozone Layer the Ozone Layer
NASA “Tot al Ozone Mapping Spect romet er” NASA “Tot al Ozone Mapping Spect romet er” http://toms. http://toms.gsfc gsfc. .nasa nasa. .gov gov/ /

SLIDE 2 Levels of Atmospheric Ozone have been Dropping Levels of Atmospheric Ozone have been Dropping
EPA EPA -
ht t p:/ / www.epa.gov/ docs/ ozone/ science/ arosa.ht ml

SLIDE 3 Decreasing Level of atmospheric ozone is harmf ul Decreasing Level of atmospheric ozone is harmf ul There has been an increase in the number There has been an increase in the number
- f cases of skin cancer and cataracts
- f cases of skin cancer and cataracts
Evidence of damage to plant and marine lif e Evidence of damage to plant and marine lif e

SLIDE 4
What is ozone? What is ozone? Where in the atmosphere is it f ound? Where in the atmosphere is it f ound? What is its purpose in the atmosphere? What is its purpose in the atmosphere? What is its chemistry? What is its chemistry? Why are levels of atmospheric ozone dropping? Why are levels of atmospheric ozone dropping? Finally, what is the Ozone Hole? Finally, what is the Ozone Hole?

SLIDE 5
Structure of Ozone Structure of Ozone
O O3
3
O Atoms O Atoms

SLIDE 6 Where is ozone f ound in the atmosphere ? Where is ozone f ound in the atmosphere ?
NASA NASA Goddard Goddard Space Flight Cent er Space Flight Cent er
Note, higher concentration in stratosphere, compared Note, higher concentration in stratosphere, compared with troposphere with troposphere

SLIDE 7 Solar Flux Solar Flux
Chemical Kinetics and Photochemical Data f or Use in Chemical Kinetics and Photochemical Data f or Use in Stratospheric Modeling Stratospheric Modeling -
JPL Publication97-
4
Role of Ozone Role of Ozone

SLIDE 8
Role of Ozone Role of Ozone Absorption Spectrum of Ozone Absorption Spectrum of Ozone

SLIDE 9 Role of Ozone Role of Ozone
“The Ozone Depletion Phenomenon”, Beyond Discovery, “The Ozone Depletion Phenomenon”, Beyond Discovery, National Academy of Sciences National Academy of Sciences

SLIDE 10
UV A (~400 to 350 UV A (~400 to 350 nm nm) not absorbed by earth’s ) not absorbed by earth’s atmosphere atmosphere Role of Ozone Role of Ozone UV B (~ 350 to 270 UV B (~ 350 to 270 nm nm) partially absorbed by ) partially absorbed by earth’s atmosphere earth’s atmosphere UV C (~270 to 150 UV C (~270 to 150 nm nm) completely absorbed by ) completely absorbed by earth’s atmosphere earth’s atmosphere UV B is harmf ul to lif e on earth UV B is harmf ul to lif e on earth

SLIDE 11 Chapman mechanism Chapman mechanism How is ozone production and destruction? How is ozone production and destruction? O O2
2 +
+ h hν ν ν ν ν ν ν ν -
> O + O O + O O + O2
2 + M
+ M-
> O3
3 + M
+ M O O3
3 +
+ h hν ν ν ν ν ν ν ν′ ′ ′ ′ ′ ′ ′ ′ -
> O + O2
2
O + O O + O3
3 -
> 2 O2
2
“Ozone: What is it and why do we care “Ozone: What is it and why do we care about it?”, NASA Facts, about it?”, NASA Facts, Goddard Goddard Space Space Flight Center Flight Center

SLIDE 12 Kinetics of Chapman Mechanism Kinetics of Chapman Mechanism Rate of f ormation of O and O Rate of f ormation of O and O3
3
d[O]/ d[O]/ dt dt = 2k = 2k1
1[O
[O2
2]
]-
k2
2[O][O
[O][O2
2][M] + k
][M] + k3
3[O
[O3
3]
] -
k4
4[O][O
[O][O3
3]
] d[O d[O3
3]/
]/ dt dt = k = k2
2[O][O
[O][O2
2][M]
][M] -
k3
3[O
[O3
3]
]-
k4
4[O][O
[O][O3
3]
] Steady Steady-
State Approximation d[O]/ d[O]/ dt dt = d[O = d[O3
3]/
]/ dt dt = 0 = 0 O O2
2 +
+ h hν ν ν ν ν ν ν ν -
> O + O k k1
1
O + O O + O2
2 + M
+ M-
> O3
3 + M
+ M k k2
2
O O3
3 +
+ h hν ν ν ν ν ν ν ν′ ′ ′ ′ ′ ′ ′ ′ -
> O + O2
2
k k3
3
O + O O + O3
3 -
> 2 O2
2
k k4
4

SLIDE 13 Kinetics of Chapman Mechanism Kinetics of Chapman Mechanism d[O d[O3
3]/
]/ dt dt = k = k2
2[O][O
[O][O2
2][M]
][M] -
k3
3[O
[O3
3]
]-
k4
4[O]
[O] [O [O3
3]
]=0 =0
k k2
2[O][O
[O][O2
2][M]/ { k
][M]/ { k3
3+k
+k4
4[O] } =
[O] } = [O [O3
3]
]
k k2
2[O][O
[O][O2
2][M]={ k
][M]={ k3
3+k
+k4
4[O] }
[O] } [O [O3
3]
]

SLIDE 14 Kinetics of Chapman Mechanism Kinetics of Chapman Mechanism Can re Can re-
write [O3
3] as:
] as:
[O3] = k2[O2][M]/ k 4 k3 /(k4[O]) +1
[O [O3
3]
] = k = k2
2[O][O
[O][O2
2][M]/ { k
][M]/ { k3
3+
+k k4
4[O]
[O] } }
(Divide by (Divide by k k4
4[O]
[O] ) )

SLIDE 15
Kinetics of Chapman Mechanism Kinetics of Chapman Mechanism Since the rate constants and concentration of Since the rate constants and concentration of species are known, can show that: species are known, can show that:
k 3 k4[O] >> 1 [O3] ≈ k 2[O2 ][M][O] k3
Hence, Hence,
[O3] = k2[O2][M]/ k 4 k3 /(k4[O]) +1
+ +

SLIDE 16 [O [O3
3] depends on rate of reaction 2
] depends on rate of reaction 2 and the intensity of light (k and the intensity of light (k3
3)
) Kinetics of Chapman Mechanism Kinetics of Chapman Mechanism
[O3] ≈ k 2[O2 ][M][O] k3
Reaction 2 is slow ( Reaction 2 is slow (termolecular termolecular); makes ozone ); makes ozone “vulnerable” to ozone “vulnerable” to ozone-
depleting reactions O O2
2 +
+ h hν ν ν ν ν ν ν ν -
> O + O k k1
1
O + O O + O2
2 + M
+ M-
> O3
3 + M
+ M k k2
2
O O3
3 +
+ h hν ν ν ν ν ν ν ν′ ′ ′ ′ ′ ′ ′ ′ -
> O + O2
2
k k3
3
O + O O + O3
3 -
> 2 O2
2
k k4
4

SLIDE 17 Competing Reactions Competing Reactions HO HOx
x cycle
cycle H, OH and HO H, OH and HO2
2 species f ormed by
species f ormed by reaction of excited O atoms with H reaction of excited O atoms with H-
- containing atmospheric species like H
containing atmospheric species like H2
2O
O and CH and CH2
2
O O3
3 +
+ h hν ν ν ν ν ν ν ν′ ′ ′ ′ ′ ′ ′ ′ -
> O + O2
2
O + H O + H2
2O
O -
> OH + OH O + CH O + CH4
4 -
> CH3
3 + OH
+ OH H H2
2O +
O + h hν ν ν ν ν ν ν ν -
> H + OH

SLIDE 18 Reactions of Reactions of HO HOx
x species with O
species with O3
3
OH + O OH + O3
3 -
> HO2
2 + O
+ O2
2
HO HO2
2 + O
+ O -
> OH + O2
2
Net Reaction Net Reaction
O + O O + O3
3 -
> 2O2
2 “Ozone Depletion” “Ozone Depletion”

SLIDE 19 NO NOx
x Cycle
Cycle Competing Reactions Competing Reactions NOx NOx species are produced during the species are produced during the reaction of O atoms with N reaction of O atoms with N 2
2O (produced in
O (produced in the soil by bacteria) the soil by bacteria) O + N O + N 2
2O
O -
> 2 NO

SLIDE 20 Reactions of Reactions of NO NOx
x species with O
species with O3
3
NO + O NO + O3
3 -
> NO2
2 + O
+ O2
2
NO NO2
2 + O
+ O -
> NO + O2
2
Net Reaction Net Reaction
O + O O + O3
3 -
> 2O2
2 “Ozone Depletion” “Ozone Depletion”

SLIDE 21 Competing Reactions Competing Reactions ClO ClOx
x cycle
cycle ClO ClOx
x species are produced f rom
species are produced f rom chlorof luorocarbons chlorof luorocarbons ( (CFC’s CFC’s) and methyl chloride ) and methyl chloride CFC’s CFC’s are artif icially produced; methyl chloride is are artif icially produced; methyl chloride is a naturally a naturally occuring
chemical. Examples of Examples of CFC’s CFC’s : : Freons Freons (CFCl (CFCl3
3, CF
, CF2
2Cl
Cl2
2)
) CCl CCl2
2F
F2
2 +
+ h hν ν ν ν ν ν ν ν -
> CF2
2Cl +
Cl + Cl Cl CCl CCl2
2F
F2
2 + O
+ O -
> CF2
2Cl +
Cl + ClO ClO

SLIDE 22 Reactions of Reactions of ClO ClOx
x species with O
species with O3
3
Cl Cl + O + O3
3 -
> ClO ClO + O + O2
2
ClO ClO + O + O -
> Cl Cl + O + O2
2
Net Reaction Net Reaction
O + O O + O3
3 -
> 2O2
2 “Ozone Depletion” “Ozone Depletion” 1995 Nobel Prize in Chemistry 1995 Nobel Prize in Chemistry

SLIDE 23 Consequences of Competing Reactions Consequences of Competing Reactions Catalytic Reactions Catalytic Reactions Cl Cl + O + O3
3 -
> ClO ClO + O + O2
2
ClO ClO + O + O -
> Cl Cl + O + O2
2
lower activation energy E Ea
a f or Chapman mechanism = 17. 1 kJ/
f or Chapman mechanism = 17. 1 kJ/ mol mol E Ea
a f or
f or ClO ClOx
x reaction = 2. 1 kJ/
reaction = 2. 1 kJ/ mol mol
catalyst catalyst catalyst catalyst intermediate intermediate intermediate intermediate

SLIDE 24 Depleting reactions are NOT independent of each Depleting reactions are NOT independent of each
- ther; in f act all occur simultaneously
- ther; in f act all occur simultaneously
Ef f ect of competing reaction on rate of ozone f ormation Ef f ect of competing reaction on rate of ozone f ormation Consequences of Competing Reactions Consequences of Competing Reactions NET LOSS OF OZONE NET LOSS OF OZONE

SLIDE 25 Sources of ozone depleting molecules Sources of ozone depleting molecules Naturally Naturally occuring
species (H2
2O, N
O, N 2
2O, CH
O, CH4
4)
) Artif icial, “man Artif icial, “man-
made” species CFC’s CFC’s (CCl (CCl3
3F, CCl
F, CCl2
2F
F2
2, etc. )
, etc. ) CCl CCl4
4, CHCl
, CHCl3
3
HBFC (CHFBr HBFC (CHFBr 2
2, CHF
, CHF2
2Br)
Br) CH CH3
3Br
Br NO f rom supersonic NO f rom supersonic aircraf ts aircraf ts The artif icial compounds have the most severe ef f ect The artif icial compounds have the most severe ef f ect

SLIDE 26 What is the “Ozone Hole”? What is the “Ozone Hole”? Every year, in October, a huge “hole” in atmospheric Every year, in October, a huge “hole” in atmospheric levels of ozone is observed over the Antarctic. levels of ozone is observed over the Antarctic.
August 1 ‘96 August 1 ‘96 -
Dec 15 ‘96
NASA NASA
Goddard Goddard Space Flight Center Space Flight Center
You will You will have to have to wait to wait to see this see this movie in movie in class. class.

SLIDE 27
Why does the Ozone Hole f orm over the Antarctic Why does the Ozone Hole f orm over the Antarctic and why in spring? and why in spring? The Antarctic Vortex The Antarctic Vortex Polar Stratospheric Clouds Polar Stratospheric Clouds Concentrations of Active Chlorine Concentrations of Active Chlorine

SLIDE 28 The Antarctic Vortex The Antarctic Vortex I n the winter, the air around the S. Pole cools I n the winter, the air around the S. Pole cools and circulate west creating a “vortex” and circulate west creating a “vortex” Air is trapped in the vortex along with Air is trapped in the vortex along with
- zone depleting species
- zone depleting species
Heat f rom outside is “shut of f ”, prolonging the Heat f rom outside is “shut of f ”, prolonging the duration of low stratospheric temperatures. duration of low stratospheric temperatures.

SLIDE 29 Polar Stratospheric Clouds Polar Stratospheric Clouds Low stratospheric temperatures result in “ice Low stratospheric temperatures result in “ice clouds” called Polar clouds” called Polar Stratopsheric Stratopsheric Clouds Clouds The surf ace of the ice clouds serve as The surf ace of the ice clouds serve as reaction sites f or heterogeneous gas reaction sites f or heterogeneous gas-
surf ace reactions reactions ClONO ClONO2
2 +
+ HCl HCl -
> HNO3
3 + Cl
+ Cl2
2
ClONO ClONO2
2 + H
+ H2
2O
O -
> HNO3
3 +
+ HOCl HOCl

SLIDE 30 Concentrations of Active Chlorine Concentrations of Active Chlorine The Cl The Cl2
2 and
and HOCl HOCl f ormed f ormed photodissociate photodissociate to to yield reactive yield reactive Cl Cl atoms atoms Cl Cl2
2 +
+ h hν ν ν ν ν ν ν ν -
> Cl Cl + + Cl Cl HOCl HOCl + + h hν ν ν ν ν ν ν ν -
> Cl Cl + OH + OH Cl Cl + O + O3
3 -
> ClO ClO + O + O2
2
OZONE DEPLETI ON OZONE DEPLETI ON

SLIDE 31 The Antarctic vortex traps The Antarctic vortex traps CFC’s CFC’s The low polar temperatures results in ice particles The low polar temperatures results in ice particles
- n which gas
- n which gas-
- solid reactions can occur ef f iciently
solid reactions can occur ef f iciently The same reactions in the gas phase have much The same reactions in the gas phase have much higher activation energies. The higher higher activation energies. The higher E Ea
a and low
and low temperatures result in very slow rates. temperatures result in very slow rates. The onset of spring corresponds to higher The onset of spring corresponds to higher light intensities and hence light intensities and hence photolysis photolysis of
Cl containing species containing species “I ngredients” f or the f ormation of the Ozone Hole “I ngredients” f or the f ormation of the Ozone Hole

SLIDE 32 Annual growth in the Antarctic Ozone Hole Annual growth in the Antarctic Ozone Hole
University of Cambridge “The Ozone Hole Tour” http:/ / www. atm. ch. cam. ac. uk

SLIDE 33 What is being done to reduce ozone depletion? What is being done to reduce ozone depletion? Montreal Protocol and subsequent treaties ban world Montreal Protocol and subsequent treaties ban world-
- wide usage of ozone depleting substances
wide usage of ozone depleting substances Assuming f ull compliance expect that ozone levels will Assuming f ull compliance expect that ozone levels will return to “natural” levels ~2050 return to “natural” levels ~2050
ht t p:/ / www. ht t p:/ / www.nobel nobel.se/ announcement .se/ announcement -
95/ announcement 95-
chemist ry.ht ml

SLIDE 34 Ref erences Ref erences NASA NASA Goddard Goddard Space Flight Center Space Flight Center ( (ht t p:/ / www. ht t p:/ / www.gsf c gsf c. .nasa nasa. .gov gov/ / ) ) EPA (www. EPA (www. epa epa. . gov gov) ) Center f or Atmospheric Science, Cambridge University Center f or Atmospheric Science, Cambridge University www. www.at m at m. .ch ch.cam.ac. .cam.ac.uk uk/ t our/ index.ht ml / t our/ index.ht ml Chemical Kinetics and Dynamics, Chemical Kinetics and Dynamics, Ch Ch 15, J. 15, J. Steinf eld Steinf eld, ,
- J. Francisco, W.
- J. Francisco, W. Hase
Hase