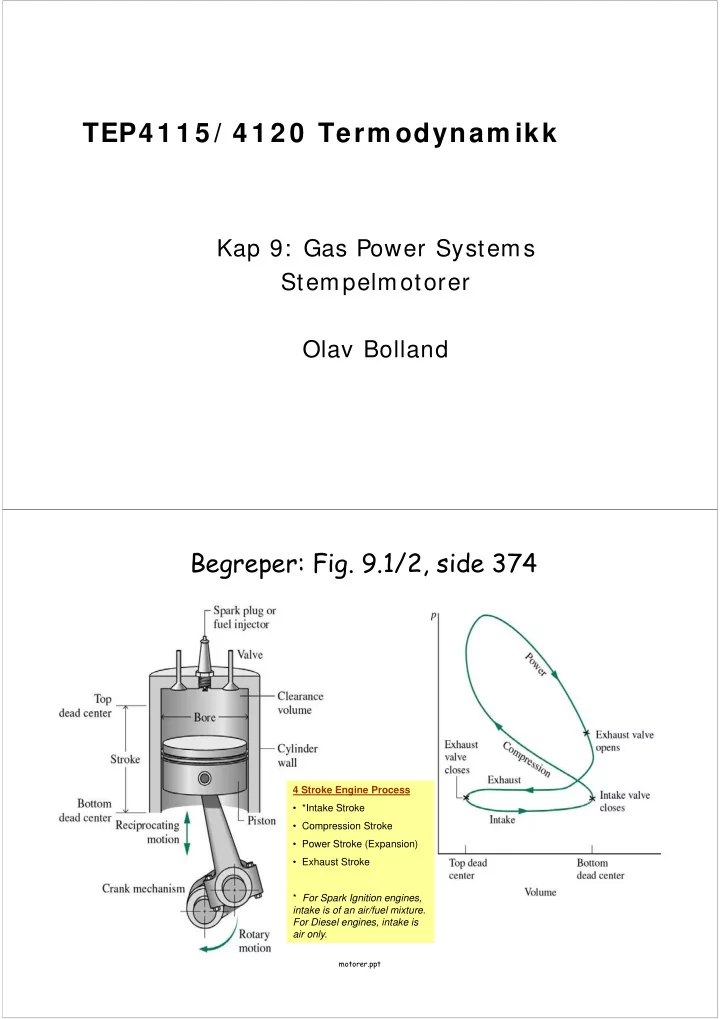
SLIDE 2 motorer.ppt
Work closed/open systems
2 1 2 1 2 2 2 1 1 1 2 2 1 2 1 1 2 1
First law for a closed system: Relating heat to entropy: Q W U U Q T dS T dS dU PdV Q T dS dU pdV U U pdV W U U W pdV
1 2 2 2 2 1 1 1 2 2 1 1 2 2 1 1
First law for an open system (steady-state): Relating heat to entropy: and and dU Q W m h m h dt Q T dS dU pdV T dS dU PdV T dS dH Vdp H U pV dU dH pdV Vdp Q dH V dp Q m dh V dp
2 2 1 1 2 1 2 1
( ) ( ) m h h V dp W m h h W V dp
motorer.ppt
Air Standard Analysis
The following assumptions are made:
- Air, an Ideal Gas, is the working fluid
- Combustion is replaced with Heat Addition
(see Chap 13 for details)
- No exhaust and intake strokes – constant
volume heat rejection
- All processes are internally reversible
For Cold-Air Standard, Specific Heats are also assumed constant