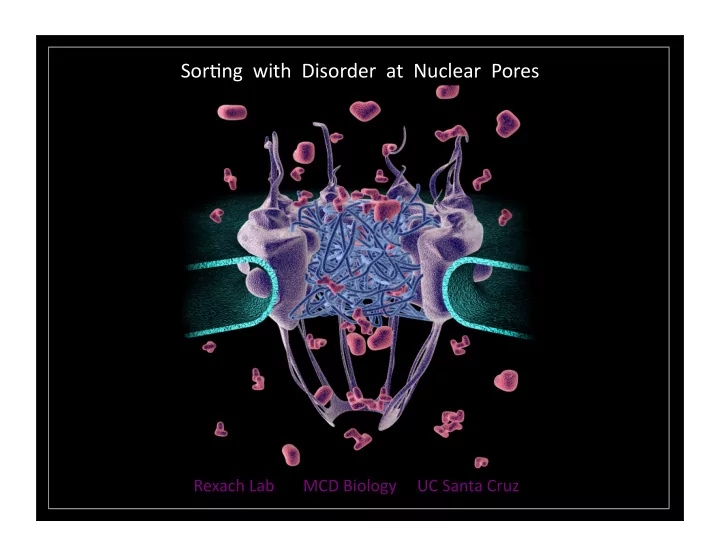
SLIDE 1
Nuclear Export ribosome subunits mRNA tRNA snRNA rRNA viral RNP’s Nuclear Import ribosomal proteins transcrip$on factors replica$on factors nucleolar proteins snRNA rRNA viral genomes
Thousands of proteins and RNP’s must cross the nuclear envelope every second
Nucleocytoplasmic traffic across the nuclear envelope
The nuclear envelope is a barrier
2