1
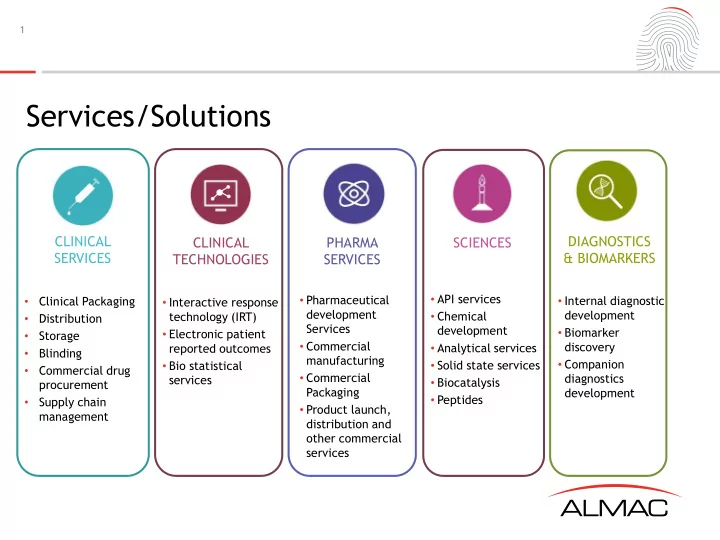
Services/Solutions
- Clinical Packaging
- Distribution
- Storage
- Blinding
- Commercial drug
procurement
- Supply chain
management
PHARMA SERVICES CLINICAL TECHNOLOGIES DIAGNOSTICS & BIOMARKERS CLINICAL SERVICES SCIENCES
- Interactive response
technology (IRT)
- Electronic patient
reported outcomes
- Bio statistical
services
- Pharmaceutical
development Services
- Commercial
manufacturing
- Commercial
Packaging
- Product launch,
distribution and
- ther commercial
services
- API services
- Chemical
development
- Analytical services
- Solid state services
- Biocatalysis
- Peptides
- Internal diagnostic
development
- Biomarker
discovery
- Companion
diagnostics development