
PRODUCTION OF PURE ENANTIOMERS AT HIGH YIELDS BY INTEGRATING - PowerPoint PPT Presentation
PRODUCTION OF PURE ENANTIOMERS AT HIGH YIELDS BY INTEGRATING CHROMATOGRAPHY, ISOMERIZATION AND MEMBRANE FILTRATION Sebastian Nimmig, Malte Kaspereit Institute of Separation Science and Technology, University Erlangen-Nrnberg PIN-NL &
PRODUCTION OF PURE ENANTIOMERS AT HIGH YIELDS BY INTEGRATING CHROMATOGRAPHY, ISOMERIZATION AND MEMBRANE FILTRATION Sebastian Nimmig, Malte Kaspereit Institute of Separation Science and Technology, University Erlangen-Nürnberg PIN-NL & NL-GUTS 9 April 2014 1
Motivation and objectives 2
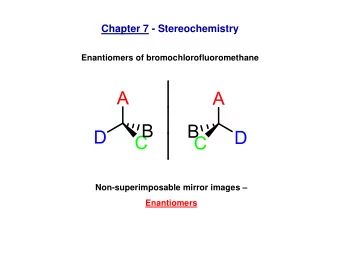
Enantiomers • Stereoisomers ("mirror images") a a C C d d b b c c • Basically identical physico-chemical properties • Often produced as racemate (50/50 mixture) • Usually only one enantiomer has the desired physiological effect Separation? Chromatography • Motivation 3
Separation of enantiomers • Yield limited to 50% only (conventional approach) E1 E1: Enantiomer 1 (product) Single-column E1+E2 E2: Enantiomer 2 Chromatography E2 Reaction required to convert E2 into E1+E2 isomerization • • Increase yield to 100% by recycling E2 solvent Single-column E1 (product) fresh feed Chromatography E2 (recycle) E1 E2 Recycle always diluted! [1] Bechtold et al ., J Biotechnology 124 (2006) 146-162 Motivation 4
• Inhibit dilution by solvent removal (here: nanofiltration) solvent Single-column E1 (product) fresh feed Chromatography E2 (recycle) Membrane- solvent E2 E1 filtration Challenges • Design specifications • Required parameters and models • Analysis and process behaviour • Fully continuous implementation • Experimental validation Objectives 5
Theoretical investigations I 6
Shortcut process design [2] • Reproduce a given chromatogram (simulated or experimental) in each cycle c 1 Concentration c / g·L -1 • Main design parameters: c 2 Feed flow rate Q feed Permeate flow rate Q perm Chrom. flow rate Q chrom Injection width Δ t in j Fractionation times t 1 , t 2 , t 3 t 1 t 1 t 1 3 =t 2 1 t 2 t 2 3 =t 3 1 t 3 t 3 3 =t k+1 1 2 2 2 1 Time t / min [2] Nimmig, Kaspereit, Chem Eng Process 67 (2013) 89– 98 Theoretical investigations I 7
Shortcut process design [2] C • Simple explicit equations R M • Easy performance prediction SC / L·g -1 100 2.5 50 Productivity PR / g·h -1 ·L -1 Shortcut R=1 80 Shortcut R<1 2 40 Yield Y / % Solvent consumption 60 1.5 30 40 1 20 20 0.5 10 0 0 0 0 0.2 0.4 0.6 0.8 1 0 0.2 0.4 0.6 0.8 1 0 0.2 0.4 0.6 0.8 1 Membrane rejection R / - Membrane rejection R / - Membrane rejection R / - Theoretical investigations I 8
Detailed process design Chromatography : Equilibrium dispersive model C Reaction: R First order kinetics, CSTR Nanofiltration: M Simplified solution diffusion model • Implementation in MatLab Theoretical investigations I 9
Detailed process design • Fully continous connection • Performance prediction: SC / L·g -1 100 80 Productivity PR / g·h -1 ·L -1 40mL 2 120mL 80 60 200mL 1.5 Yield Y / % Solvent consumption 60 280mL 40 360mL 1 40 440mL 20 0.5 20 0 0 0 0 0.2 0.4 0.6 0.8 1 0 0.2 0.4 0.6 0.8 1 0 0.2 0.4 0.6 0.8 1 Membrane rejection R / - Membrane rejection R / - Membrane rejection R / - Theoretical investigations I 10
Experimental validation 11
C UV UV PI PI R TI F TI F M Experimental validation 12
Experiment 1 – Design via detailed simulation 0.1 Simulation Concentration c out / g·L -1 1 Concentration c / g·L -1 Experiment 1 0.08 0.1 0.06 0.04 0.05 0.02 0 0 15 20 25 30 35 5 10 15 20 Time t / min Fraction # 100 Simulation Concentration c out / g·L -1 1 0.2 Experiment 1 Purity PUR / % 95 0.15 90 0.1 85 0.05 0 80 0 100 200 300 0 5 10 15 20 Time t / min Fraction # Experimental validation 13
Experiment 2 – Design via shortcut method 0.25 0.3 Design Concentration c out / g·L -1 Concentration c / g·L -1 Experiment 2 0.2 0.2 0.15 0.1 0.1 0.05 0 0 20 30 40 0 5 10 15 20 Time t / min Fraction # 0.4 100 Experiment 2 Concentration c out / g·L -1 1 90 0.3 Purity PUR / % 80 0.2 70 0.1 60 0 50 0 100 200 300 0 0 5 10 15 20 Time t / min Fraction # Experimental validation 14
Theoretical investigations II 15
Unit Arrangements Different setups - different performance? 1 2 3 C C C M R M R M R 4 5 6 C C C R M R M R M R: Reactor C: Column M: Membrane : Fresh Feed Theoretical investigations II 16
Perfomance prediction - Yield Da = 0.3 Da = 10 100 100 90 90 Best choice: 80 80 4 70 70 C Yield Y / % Yield Y / % 60 60 R M 50 50 40 40 30 30 20 20 0.4 0.6 0.8 1 0.4 0.6 0.8 1 Membrane rejection R / - Membrane rejection R / - Var 1 Var 3 Var 5 Batch Var 2 Var 4 Var 6 Theoretical investigations II 17
Perfomance prediction - Productivity Da = 0.3 Da = 10 3 3 Best choice: Productivity PR / g·h -1 ·L -1 Productivity PR / g·h -1 ·L -1 4 2.5 2.5 C R M 2 2 1.5 1.5 0.4 0.6 0.8 1 0.4 0.6 0.8 1 Membrane rejection R / - Membrane rejection R / - Var 1 Var 3 Var 5 Batch Var 2 Var 4 Var 6 Theoretical investigations II 18
Further extensions of concept? 19
heating reaction jacket chamber stirrer membrane Further extensions of concept 20
On-column protein refolding SolvR N Single-column Native protein D Chromatography X ( N ) Undesired Membrane reactor conformations SolvD ( X ) D X Denatured protein ( D ) Solvent Further extensions of concept 21
Summary • Proposed concept capable of significantly improving yield and performance • Shortcut methods developed for basic design, full model for detailed design • Performance limited mainly by membrane rejection • Process setup influences performance • First successful realization of such process in directly coupled operation Outlook • Potential application to industrial relevant compunds? Summary 22
Thank you for your attention! 23
Chlorthaidone racemization • Nearly insolubile in Water • Increasing solubility in MeOH/H 2 O • MW =338 g/mol • Kinetics known as function of pH-value and temperature [3] [3] J. G. Palacios, B. Kramer, A. Kienle, M. Kaspereit, J Chromatogr A 1218 (2011) 2232- 2239 Racemization under acid conditions Racemization scheme 24
Reactor concentration behavior for different Volumes V R =1mL V R =1000mL Detailed process design 25
I B F E P • Membran Reactor design I N Reaktor Membranmodul • Membrane unit acts as CSTR (residence time function) Membran behavior 26
Parameter determination - Nanofiltration Pure water experiments (k1) Batch concentration experiments (k2) 500 600 8.30 bar 450 550 6.38 bar 2 ] Permeate flux for pure solvent [mL/min/m 4.80 bar 400 500 2 ] Permeate flux [mL/min/m 350 450 300 400 250 350 200 300 150 250 100 200 50 150 0 100 0 2 4 6 8 0 0.1 0.2 0.3 0.4 Pressure ∆ p [bar] Concentration c CTD [g/L] Experimental characterization 27
• Parameter determination - Racemization Concentration [g/L] ln(c*) Time [min] Time [min] Experimental characterization 28
Recommend
More recommend
Explore More Topics
Stay informed with curated content and fresh updates.










![[12] The Eigenvector Two interest-bearing accounts Suppose Account 1 yields 5% interest and](https://c.sambuz.com/718883/12-the-eigenvector-two-interest-bearing-accounts-s.webp)










