SLIDE 1
Chapter 7 - Stereochemistry Enantiomers of bromochlorofluoromethane - - PowerPoint PPT Presentation
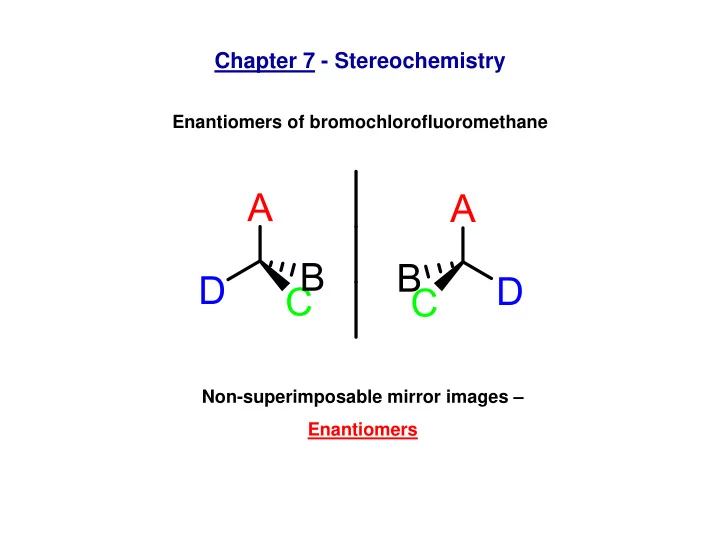
Chapter 7 - Stereochemistry Enantiomers of bromochlorofluoromethane - - PowerPoint PPT Presentation
Chapter 7 - Stereochemistry Enantiomers of bromochlorofluoromethane Non-superimposable mirror images Enantiomers Figure 7.1 7.12 Optically active molecules of biological importance O O O N OH N O N NH 2 O N H HO H codeine
SLIDE 2
SLIDE 3
O O HO H N codeine O N N O strychnine HO H H H cholesterol NH2 N H O OH tryptophan NH2 HO HO L-dopa OH OH HO O HO O OH OH HO HO O sucrose
7.12 Optically active molecules of biological importance
SLIDE 4
$100 billion sales worldwide in 2000 Account for 32% of the $360 billion total drug sales
SLIDE 5
7.2 The Chirality Center
Carbon atom is asymmetric C is a stereogenic center Enantiomers are stereoisomers since the atoms at the stereogenic carbon are arranged differently in space.
SLIDE 6
Mirror images of chlorodifluoromethane are superimposable Figure 7.2
Achiral i.e. not chiral
7.3 Symmetry in achiral structures
SLIDE 7
7.4 Optical activity
Figure 7.4 Typical polarimeter setup : [α]D = 100 x (rotation)/(cell length) x (concentration)
SLIDE 8
7.8 Enantiomers
same physical properties except rotation of plane polarized light
- ne enantiomer positive rotation (+) other negative rotation (-)
SLIDE 9
Which molecules contain chiral (stereogenic) centers?
The stereogenic C must have 4 different groups attached
SLIDE 10
7.5 Absolute and Relative Configuration
Absolute Configuration – Actual arrangement of substituents in space (+)-2-butanol and (-)-2-butanol, but which is which? Relative Configuration - Configuration relative to another compound. Pre-1951, compounds could be related to each other but the absolute configuration was not able to be determined.
SLIDE 11
7.6 Nomenclature - Use of the Cahn-Ingold-Prelog System (R) and (S)
S enantiomer R enantiomer R - Rectus - the clockwise arrangement of groups S - Sinestre - the counterclockwise arrangement of groups
SLIDE 12
7.6 Nomenclature - Use of the Cahn-Ingold-Prelog System
SLIDE 13
7.7 Fischer projection formulas
Figure 7.5
SLIDE 14
7.9 Reactions that create a Chirality Center
Figure 7.6
SLIDE 15
7.10 Chiral molecules with two Chirality Centers
Figure 7.7
SLIDE 16
7.10 Representations of (2R, 3R)-dihydroxybutanoic acid
Figure 7.8
All the same molecule: (a) and (b) differ only by bond rotation (b) leads to correct Fischer projection Conversion of “zig-zag” picture to Fischer projection
SLIDE 17
7.10 Chiral molecules with two Chirality Centers
SLIDE 18
7.11 Achiral molecules with two Chirality Centers
Figure 7.9
SLIDE 19
Meso-2,3-butanediol
Figure 7.10
SLIDE 20
7.12 Stereogenic centers in cholic acid
Figure 7.11
SLIDE 21
O O HO H N codeine O N N O strychnine HO H H H cholesterol NH2 N H O OH tryptophan NH2 HO HO L-dopa OH OH HO O HO O OH OH HO HO O sucrose
7.12 Optically active molecules of biological importance
SLIDE 22
7.13 Reactions that produce diastereomers
Figure 7.12
SLIDE 23
7.14 Resolution of a chiral substance into its enantiomers
Figure 7.13
SLIDE 24