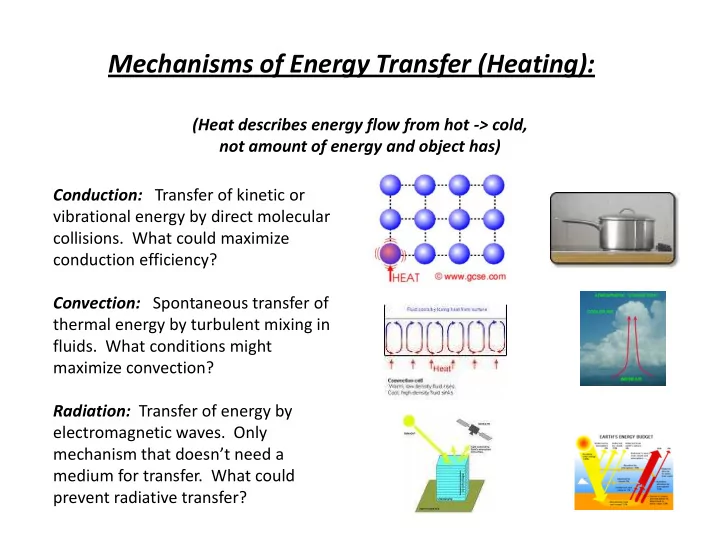
Mechanisms of Energy Transfer (Heating):
(Heat describes energy flow from hot -> cold, not amount of energy and object has) Conduction: Transfer of kinetic or vibrational energy by direct molecular
- collisions. What could maximize
conduction efficiency? Convection: Spontaneous transfer of thermal energy by turbulent mixing in
- fluids. What conditions might