SLIDE 4 Raltegravir Raltegravir (MK 0518): (MK 0518): A Novel HIV A Novel HIV-
1 Integrase Inhibitor
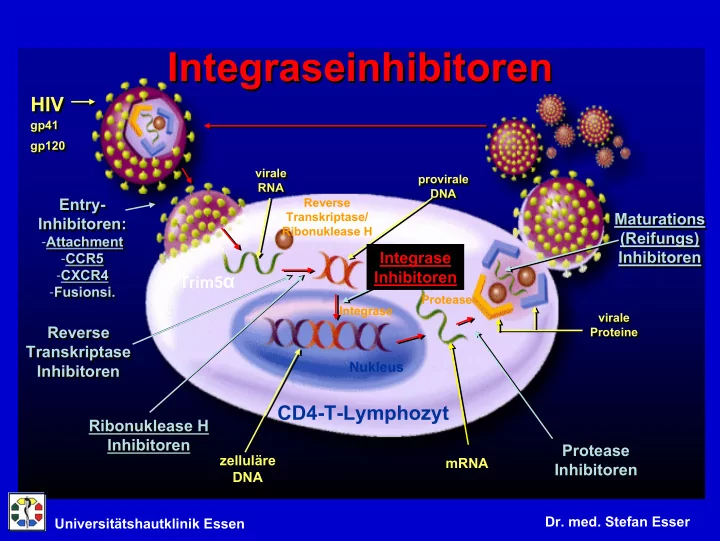
- A new mechanism of action:
A new mechanism of action: HIV-1 Integrase Transfer Inhibitor
Potent in vitro in vitro activity activity
IC95
95 = 33 nM
= 33 nM ± ± 23 nM in 50% human serum 23 nM in 50% human serum
Active against:
– – multi multi-
drug resistant HIV-
1 – – CCR5 and CXCR4 HIV CCR5 and CXCR4 HIV-
1
- HIV resistant to raltegravir remain
HIV resistant to raltegravir remain sensitive to other ARTs sensitive to other ARTs
Synergistic in vitro in vitro with all ARTs tested with all ARTs tested
- Potent activity in combination therapy in Phase II studies
Potent activity in combination therapy in Phase II studies – – in ART in ART-
naive patients at Week 24 at Week 24 (Markowitz et al, IAC 2006, Abst
(Markowitz et al, IAC 2006, Abst THLB0214) THLB0214)
85 – – 95% with HIV RNA < 50 copies/mL 95% with HIV RNA < 50 copies/mL – – In patients failing therapy with triple class resistant virus at In patients failing therapy with triple class resistant virus at Week Week 24 24 (Grinsztejn et al, ICAAC 2006, Abst H
(Grinsztejn et al, ICAAC 2006, Abst H-
1670b)
57-
67% with HIV RNA < 50 copies/mL with HIV RNA < 50 copies/mL
N N H N O O- O H N F O N N O K+
- D. Cooper, R. Steigbigel et al., 14th CROI Los Angeles 2007, Abstract # 105 a&b LB
Universitätshautklinik Essen