SLIDE 1
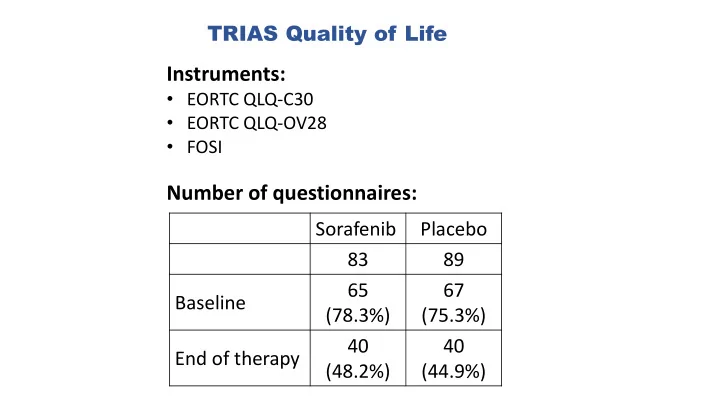
TRIAS Quality of Life
Instruments:
- EORTC QLQ-C30
- EORTC QLQ-OV28
- FOSI
Instruments: EORTC QLQ-C30 EORTC QLQ-OV28 FOSI Number of - - PowerPoint PPT Presentation
TRIAS Quality of Life Instruments: EORTC QLQ-C30 EORTC QLQ-OV28 FOSI Number of questionnaires: Sorafenib Placebo 83 89 65 67 Baseline (78.3%) (75.3%) 40 40 End of therapy (48.2%) (44.9%) TRIAS Quality of Life EORTC
5 10 15
10 20 30
10 20 30
10 20
▬ 95%