SLIDE 1
HP 5988A EI/CI GC/DIP/MS System uses a quadrupole mass analyzer - - PowerPoint PPT Presentation
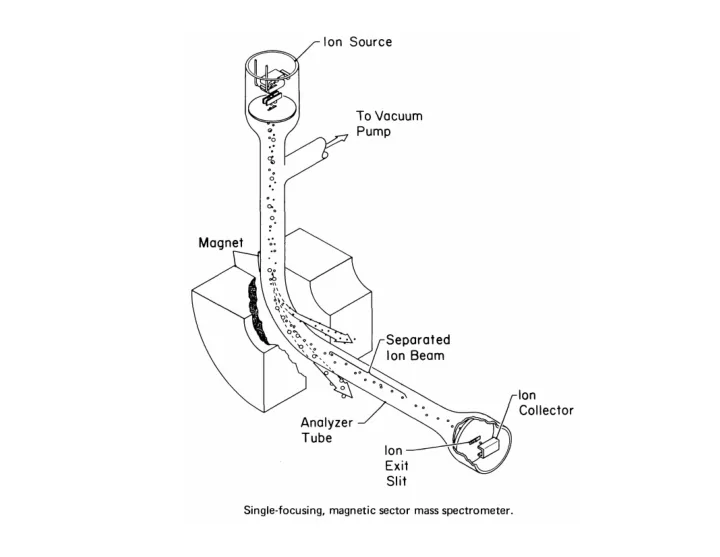
HP 5988A EI/CI GC/DIP/MS System uses a quadrupole mass analyzer - - PowerPoint PPT Presentation
HP 5988A EI/CI GC/DIP/MS System uses a quadrupole mass analyzer Agilent 6850/5973 GC/MSD uses a quadrupole analyzer ABI Voyager Pro MALDI-TOF MS system Mass spectrometers weight individual molecules and pieces of individual molecules. Mass
SLIDE 2
SLIDE 3
Agilent 6850/5973 GC/MSD uses a quadrupole analyzer
SLIDE 4
ABI Voyager Pro MALDI-TOF MS system
SLIDE 5
Mass spectrometers weight individual molecules and pieces of individual molecules. Mass spectrometry is useful for
- Determining molecular mass
< at unit resolution for example, benzene has MF C6H6 MW = 6(12) + 6(1) = 78 g/mol in mass spectrum, observe m/z M+C = 78 amu
- Determining molecular formula
< at unit resolution isotopic contributions to (M+1)+C and (M+2)+C allow MF to be deduced < at high resolution mass spectral resolution of <5 ppm allows MF to be identified
- Determining structure
< the gas-phase elimination and rearrangement mechanisms of M+C are diagnostic of constitution
SLIDE 6
N O O O O
300 280 260 240 220 200 180 160 140 120 100 80 60 40 20 8 16 24 32 40 48 56 64 72 80 88 96 303 272 198 182 152 105 82 51 42
Cocaine C17H21NO4 m/z = 303
SLIDE 7
ION Height 303 M+C 100
- 204
C17 (M+1)+C 19.7 99 (M+2)+C 0.8
- 14
N 85 maximum # C in MF: 1.1x # 20
- 64
O4 x # 18 21
- 21
H21 if C17, contribution to (M+1)+C = 1.1(17) = 18.7 M+C remaining (M+1)+C = 20 - 18.7 = 1.3 if C18, contribution to (M+1)+C = 1.1(18) = 19.8 remaining (M+1)+C = 20 - 19.8 = 0.2 M+C is odd, so N is in MF, have to choose C17 for C17N1 remaining (M+1)+C = 1.3 - 0.37 = 0.9 maximum #O in MF: 0.20y # 0.8 y # 4 (M+1)+C for C17NO4, remaining (M+1)+C = 0.2 - 0.2 = 0 The number of amu left corresponds to H’s (M+2)+C
C17H21NO4
relative abundances of isotopes M
12C
100
1H
100
14N
100
16O
100
35Cl
100
79Br
100 M+1
13C
1.1
2H
0.015
15N
0.37
17O
0.04
- M+2
- 18O
0.20
37Cl
32
81Br
97.3
SLIDE 8
M @
+
85 80 75 70 65 60 55 50 45 40 35 30 25 20 15 10 20 30 40 50 60 70 80 90 100 86 72 71 59 53 43 41 31 27 15
OH C5H10O 2-Methyl-3-buten-2-ol
SLIDE 9
110 100 90 80 70 60 50 40 30 20 10 20 30 40 50 60 70 80 90 100 114 99 86 81 71 58 53 43 27 15
M @
+
C7H14O O O O O O
. . .
+
. . .
+
. . . . . . .
+
m/z = 43 . . . . . . . + O O . . .
+ +
m/z = 71 + .
2-Heptanone
SLIDE 10
120 110 100 90 80 70 60 50 40 30 20 10 10 20 30 40 50 60 70 80 90 100 120 105 93 79 41 26 15
Br C3H5Br Br . . . . . . . . . . . Br .
+
+ m/z = 79
+
Br . . . . . . . . . . . Br .
+
+ m/z = 41
+
3-Bromo-1-propene
SLIDE 11
(C26H19O2)+ m/z = 363.1386 found (corrected): m/z = 363.1804 O O + H+
SLIDE 12
Calculate the exact mass of (C26H19O2)+ Element Mass C 12.00000000 26(12.00000000) + 19(1.007825035) + 2(15.99491463) H 1.007825035 O 15.99491463 = 363.1385 found (corrected) m/z = 363.1804 the difference is -.0419 amu , which corresponds to a 0.01% deviation from the calculated exact mass this is a difference of 115 ppm, very large compared to the accuracy of the measurement, which is 5 ppm MF with better matches: (C27H23O)+ 363.1749 ∆ 15 ppm (C24H27O3)+ 363.1960 ∆ 43 ppm
SLIDE 13
O O H3N N N H + (C13H16N3O2)+ m/z = 246.1239 found (corrected): m/z = 246.1246 2.8 ppm difference (best match for this MF)
SLIDE 14
Solvolysis of 3-bromo-3-methyl-1-butyne (M.W. 147) in water at 50 EC for 12 hours gives two products, A and B. The infrared and mass spectra
- f product A are shown below:
IR spectrum A Mass spectrum A
SLIDE 15
Solvolysis of 3-bromo-3-methyl-1-butyne (M.W. 147) in water at 50 EC for 12 hours gives two products, A and B. The infrared and mass spectra
- f product A are shown below:
2100 cm-1 C/ / / /C stretch 3300 cm-1 Csp–H stretch < 3000 cm-1: Csp3–H stretch > 3000 cm-1: ~1600 cm-1 Csp2–Hstretch C=C stretch 910 cm-1 625 cm-1 R2C=CH2 bend RC/ / / /CH bend Mass spectrum A Clearly label two features on the IR spectrum indicating the functional group class of product A.
SLIDE 16
Solvolysis of 3-bromo-3-methyl-1-butyne (M.W. 147) in water at 50 EC for 12 hours gives two products, A and B. The infrared and mass spectra
- f product A are shown below:
2100 cm-1 C/ / / /C stretch 3300 cm-1 Csp–H stretch < 3000 cm-1: Csp3–H stretch > 3000 cm-1: ~1600 cm-1 Csp2–Hstretch C=C stretch 910 cm-1 625 cm-1 R2C=CH2 bend RC/ / / /CH bend Mass spectrum A M+ A A A A Clearly label the molecular ion (M+ A)
- f the mass spectrum.
SLIDE 17
Solvolysis of 3-bromo-3-methyl-1-butyne (M.W. 147) in water at 50 EC for 12 hours gives two products, A and B. The infrared and mass spectra
- f product A are shown below:
2100 cm-1 C/ / / /C stretch 3300 cm-1 Csp–H stretch < 3000 cm-1: Csp3–H stretch > 3000 cm-1: ~1600 cm-1 Csp2–Hstretch C=C stretch 910 cm-1 625 cm-1 R2C=CH2 bend RC/ / / /CH bend Mass spectrum A M+ A A A A The fragment appearing at mass 51 suggests a loss of what functional group from the molecular ion? 66 - 51 = 15 mass units: C (12) + 3 [H (1)] = CH3 (15)
SLIDE 18
Solvolysis of 3-bromo-3-methyl-1-butyne (M.W. 147) in water at 50 EC for 12 hours gives two products, A and B. The infrared and mass spectra
- f product A are shown below:
2100 cm-1 C/ / / /C stretch 3300 cm-1 Csp–H stretch < 3000 cm-1: Csp3–H stretch > 3000 cm-1: ~1600 cm-1 Csp2–Hstretch C=C stretch 910 cm-1 625 cm-1 R2C=CH2 bend RC/ / / /CH bend Mass spectrum A M+ A A A A Provide a structural formula for product A.
SLIDE 19
Solvolysis of 3-bromo-3-methyl-1-butyne (M.W. 147) in water at 50 EC for 12 hours gives two products, A and B. The infrared and mass spectra
- f product A are shown below: