SLIDE 1
CO– SiC Si
- O
Si
GENERATION OF SILYLETHYNOLATES FROM DISILYLKETENES
- O
Si – Nu– transmetallation addition Si Si O– Nu – SiNu Me3Si
- O
SiMe3 Nu THF HMPA t-BuMe2Si
- O
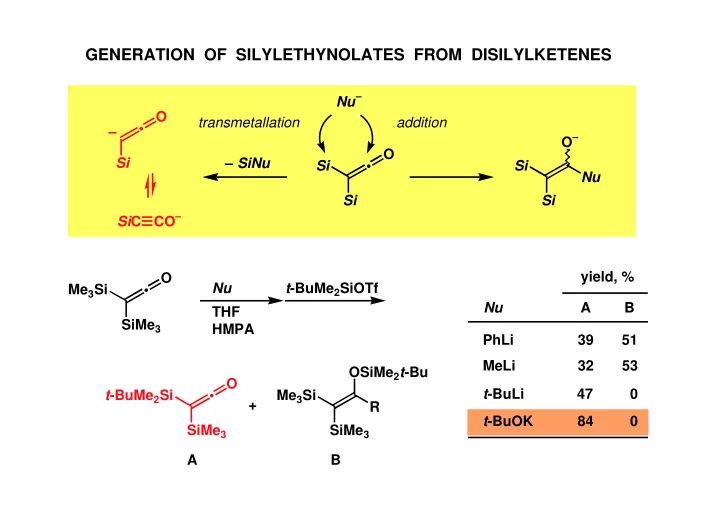
GENERATION OF SILYLETHYNOLATES FROM DISILYLKETENES Nu O - - PowerPoint PPT Presentation
GENERATION OF SILYLETHYNOLATES FROM DISILYLKETENES Nu O transmetallation addition O O Si SiNu Si Si Nu Si Si CO Si C yield, % O t -BuMe 2 SiOTf Nu Me 3 Si A B Nu THF SiMe 3 HMPA PhLi 39 51