SLIDE 1
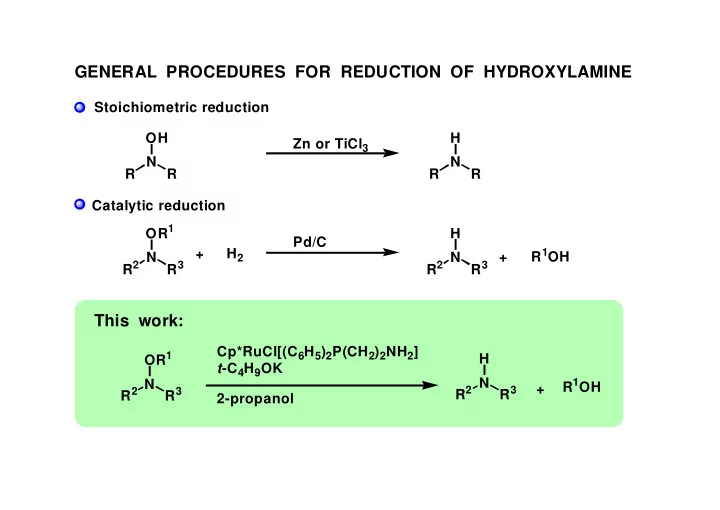
GENERAL PROCEDURES FOR REDUCTION OF HYDROXYLAMINE
Stoichiometric reduction Catalytic reduction R N R OH Zn or TiCl3 R N R H R2 N R3 OR1 R2 N R3 H Pd/C + H2
This work:
Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK 2-propanol R2 N R3 OR1 R2 N R3 H + R1OH + R1OH

SLIDE 2 sel, % >99 83 68 86 68 57
LIGAND EFFECT
Cp*RuCl(L–L) t-C4H9OK 2-propanol 80 °C, 18 h N OH N H + (C6H5)2P NH2 (C6H5)2P NHCH3 (C6H5)2P N(CH3)2 (CH3)2N NH2 Ph Ph Ph Ph NH2 N S/C=100, Ru:t-C4H9OK=1:2, [hydroxylamine] = 0.5 M aminea imineb nitrona 81 59 98 >99 98 >99 77 49 66 86 67 56 <1 2 7 7 7 8 <1 6 19 5 20 29
a Isolated yield. b Determined by 1H NMR.
conv, %a N Ph Ph N O Ph Ph + L–L yield, %

SLIDE 3
HYDROGEN SOURCE
H2 Cp*RuCl(L–L) t-C4H9OK 2-propanol 80 °C, 2 h N OH N H + L–L Ph Ph Ph Ph H2O conv, %a yield, %b H2, atm (C6H5)2P NH2 (CH3)2N NH2 5 5 75 43 71 36
a Determined by NMR. b Isolated yield.
42 41 + S/C = 100, Ru:t-C4H9OK = 1:2, [hydroxylamine] = 0.5 M 16 18 OH + O

SLIDE 4
SOLVENT EFFECT
Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK solvent–2-propanol 80 °C, 18 h N OH N H + solvent Ph Ph Ph Ph H2O CH3CN toluene conv, %a yield, %a 2-propanol S/C = 100, Ru:t-C4H9OK = 1:2, [hydroxylamine] = 0.5 M Condition: Solvent 3 ml, 2-propanol 1 ml
a Isolated yield.
32 10 81 77 44 36 sel, % >99 >99 31

SLIDE 5 EFFECT OF SUBSTITUENTS
Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK 2-propanol 80 °C, 2 h R2 N R3 OR1 R2 N R3 H + R1OH conv, %a hydroxylamine
a Determined by 1H NMR & 13C NMR. b n.d. not determined.
>99 >99 yield, %a 30 88 amine alcohol S/C = 10, Ru:t-C4H9OK = 1:2, [hydroxylamine] = 0.1 M Ph OH Ph NH2 Ph NH2 no reaction >99 74 H N Ph Ph >99 85 n.d.b Ph OH NH3 OH N H Ph NH O Ph N O Ph Ph Ph NH2 O Ph N OH Ph Ph Ph

SLIDE 6 REDUCTION OF NITRON WITH ALCOHOLS
N O Ph Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK 2-propanol, 80 °C, 18 h S/C = 100, Ru:t-C4H9OK = 1:2, [nitron] = 0.5 M Ph N Ph Ph N Ph Ph H + 95% conv. 73%a yield 4%b N Ph Ph OH 18%a
a isolated yield b determined by NMR
+
REDOX DISPROPORTIONATION OF HYDROXYLAMINE
N OH Ph Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK benzene-d6, 80 °C, 24 h S/C = 100, Ru:t-C4H9OK = 1:2, [hydroxylamine] = 0.5 M Ph N Ph Ph N Ph Ph H + 89% conv. O ca 1 : 1

SLIDE 7
O N CH3 O N >99% yield determined by GC O CH3 Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK 2-propanol, 80 °C, 18 h S/C = 100, Ru:t-C4H9OK = 1:2, [N-oxide] = 0.5 M
REDUCTION OF N-OXIDE WITH ALCOHOLS REDUCTION OF IMINE WITH ALCOHOLS
CH3 N Ph Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK 2-propanol, 30 °C, 24 h S/C = 100, Ru:t-C4H9OK = 1:2, [imine] = 0.5 M CH3 N Ph H 30% yield determined by NMR + H2O

SLIDE 8
A POSSIBLE MECHANISM
R N OH R H N R R [Ru] R N O R + + H2O [Cp*Ru(PN)] R N R [Cp*Ru(PN)]-H2 [Cp*Ru(PN)]-H2 [Cp*Ru(PN)] [Cp*Ru(PN)]-H2 [Cp*Ru(PN)] + H2O N Ru P H H H Ar Ar polar substrates δ+ δ-

SLIDE 9
SUMMARY
Cp*RuCl[(C6H5)2P(CH2)2NH2] t-C4H9OK 2-propanol R2 N R3 OR1 R2 N R3 H + R1OH Catalytic N–O single bond cleavage High chemoselectivity Reductive transformation of hydroxylamine leading to amine & alcohol