SLIDE 1
COLi R3SiC R3SiC CLi R3Si
- O
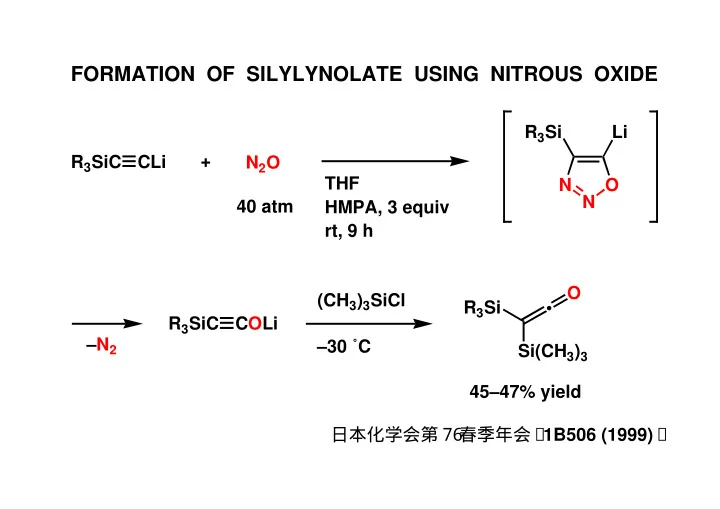
FORMATION OF SILYLYNOLATE USING NITROUS OXIDE R 3 Si Li + R 3 - - PowerPoint PPT Presentation
FORMATION OF SILYLYNOLATE USING NITROUS OXIDE R 3 Si Li + R 3 SiC CLi N 2 O THF N O N 40 atm HMPA, 3 equiv rt, 9 h O (CH 3 ) 3 SiCl R 3 Si R 3 SiC COLi N 2 30 C Si(CH 3 ) 3 4547% yield 7 6