Enzymes! Biology Intro Video https://www.youtube.com/watch?v=XTUm- - - PowerPoint PPT Presentation
Enzymes! Biology Intro Video https://www.youtube.com/watch?v=XTUm- - - PowerPoint PPT Presentation
Enzymes! Biology Intro Video https://www.youtube.com/watch?v=XTUm- 75-PL4 An enzyme is a protein that functions as a catalyst to speed up a chemical reaction in the body. Continuously recycled over and over again. Biological
Intro Video
- https://www.youtube.com/watch?v=XTUm-
75-PL4
- An enzyme is a protein that functions as a catalyst
to speed up a chemical reaction in the body.
- Continuously recycled over and over again.
- Biological catalyst: speed up rates of reactions
inside the cytoplasm.
- They control the rate of metabolic reactions in the
body.
- They lower activation energy (energy needed to
get a reaction started).
- They weaken chemical bonds so molecules can be
made or broken down by the body.
- Highly specific: catalyze only one chemical reaction
at a time and they have a specific substrate.
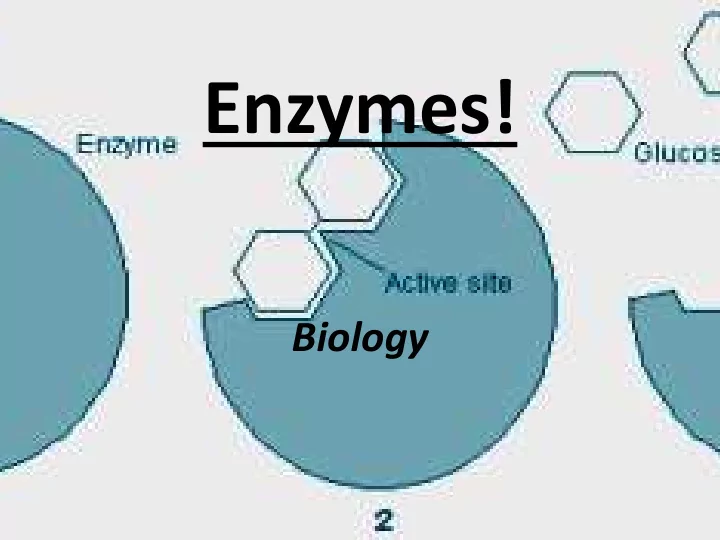
- Substrate : the reactant in the chemical reaction
that is catalyzed by the enzyme, the substance that is changed.
- Active Site: the region on the enzyme where the
substrate attaches. The shape of the active site changes based on the substrate.
- Product: what is created after the chemical
reaction has occurred.
Specific Terminology
- Many enzymes have an “ase” ending.
- A few popular ones:
– Catalase-> breaks down hydrogen peroxide in eukaryotic cells – Sucrose (table sugar) -> sucrase – Lipids-> lipase – Proteins-> Proteases – Amylase-> human saliva, helps break down starch (amylose) – Lactose -> Lactase – DNA helicase-> unzips the double stranded DNA molecule for replication – DNA polymerase-> enzymes that create DNA molecules by assembling nucleotides
Fun Fact!
- Catalase breaks down hydrogen peroxide in
eukaryotic cells.
- It’s estimated that 40 million hydrogen
peroxides go into a catalase enzyme every second.
- Hydrogen peroxide is toxic to our cells in
large quantities
https://www.youtube.com/watch?v=3Tn- 7JcZJuQ&ebc=ANyPxKroxLtgDhGJngKVMtc5s- x0rTynj- TXwGMvf4hSY5G_i7nJkJc9ampYnrF6krm- 1RzRCN8DzYTBNcZTRYSUG_AquKdc8g&spfrelo ad=10
Enzyme Substrate Complex
- Steps:
– 1. Substrate binds to the enzyme at the active site. – 2. The substrate is changed by the enzyme and converts the reactants into products. – 3. Products are released into the body. – 4. Once the products are released, the active site is ready for another molecule (substrate) to bind to. – 5. The process is ongoing, it never stops.
- This is often referred to as the “Lock and Key
Model” or the “Enzyme Substrate Complex”
- The shape of the active site (“the lock”)
determines which substrate (“the key”) will “fit” into the enzyme.
- Lowers activation energy so that the product can form
and the chemicals can spontaneously break apart.
- Picture from Biology Holt McDougal text, 2012
Factors that Affect Enzymes
- 1. Temperature:
- As temperature increases, kinetic energy
increases, and molecules move around a lot
- more. The more they move around, the higher
the probability that an enzyme and a substrate will bind together and react.
– Enzymatic reactions increase with an increase in temperature.
- Works best at a certain pH.
– Sensitive to changes in pH, especially acidity – Too low or too high, the enzyme will DENATURE (fall apart).
https://www.google.com/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ve d=0ahUKEwjv66SYoJ7LAhXMGR4KHez1CdMQjB0IBg&url=http%3A%2F%2Fwww.rsc.org%2Flearn
- chemistry%2Fresources%2Fchemistry-in-your-
cupboard%2Fvanish%2F8&psig=AFQjCNFN6wFjYVL_Vc6UFl- cb2Bf7Iu5Ag&ust=1456875515003082
- 3. Concentration of Enzyme or
Substrate
- As the enzyme
concentration increases, the rate of reaction increases.
If enzyme concentration is low, the reaction is slow.
http://www.rsc.org/Education/Teachers/Resou rces/cfb/enzymes.htm
- As the
substrate increases, the reaction increases, up to a certain point (enzyme is limited).
http://faculty.clintoncc.suny.edu/faculty/micha el.gregory/files/bio%20101/bio%20101%20lab
- ratory/enzymes/enzymes.htm
Activity / Homework
- Complete the Enzyme- Substrate
Manipulation Activity
Enzyme Substrate Complex: Graphing and Manipulative Lab
Videos
- https://www.youtube.com/watch?v=NdMVRL
4oaUo
- https://vimeo.com/86362472
ATP & Energy
Our main energy currency
- ATP= Adenosine Triphosphate
- Macromolecule: Nucleic Acid
- Organelle: Mitochondria
- Main Function: Main energy currency in all
living things.
- Comes from: breakdown of glucose
(carbohydrates).
Structure: Made up of adenine, ribose, and three phosphates
Differences
ATP ADP
3 phosphates High energy 2 phosphates Low energy
phosphate removed
- Fats store the most energy.
– 80 % of the energy in your body. – About 146 ATP molecules from a triglyceride.
- Proteins are the least likely to be broken down to
make ATP because they have so many different functions.
– Amino acids are not usually needed for energy. – About the same amount of energy as a carbohydrate.
- Carbohydrates are the molecules most commonly
broken down to make ATP.
– Not stored in large amounts. – Up to 36 ATP is created from one glucose molecule.
Of course it all starts with photosynthesis, and then organisms take it in via cellular respiration
- What if there isn’t any sunlight? Where does
the energy come from?
– Chemosynthesis: process by which organisms use chemical energy to make their food. – Where does this occur?
- Deep sea hydrothermal vents.
https://www.youtube.com/watch?v=XotF9fzo4 Vo
Review Questions
- 1. Explain in your own words, what is occurring in the ATP / ADP cycle.
- 2. Describe two functions of catalysts in chemical reactions.
- 3. The substrate is also known as the _________________ in a
chemical reaction.
- 4. List three ways in which enzymes can be altered.
- 5. Some organisms live in very hot or very acidic environments. Would
their enzymes function in a person’s cells? Why or why not?
- 6. Suppose that the amino acids that make up an enzyme’s active site
are changed, how might this change affect the enzyme?
- 7. What is the main function of ATP?
- 8. How do we obtain ATP?
- 9. Which organic molecule is used by the body as a good source of long
term energy storage?
- 10. Proteins are composed of chains of ______________________