SLIDE 1
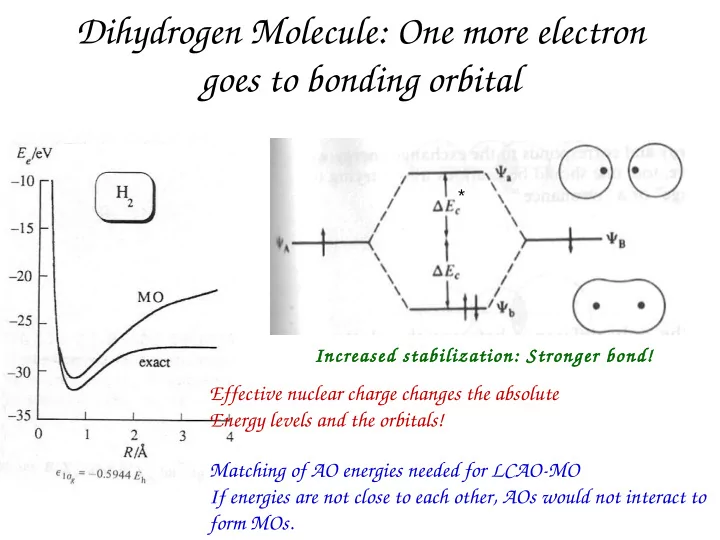
Dihydrogen Molecule: One more electron goes to bonding orbital
Increased stabilization: Stronger bond!
*
Dihydrogen Molecule: One more electron goes to bonding orbital * - - PowerPoint PPT Presentation
Dihydrogen Molecule: One more electron goes to bonding orbital * Increased stabilization: Stronger bond! Effective nuclear charge changes the absolute Energy levels and the orbitals! Matching of AO energies needed for LCAO-MO If energies are
*
He2
+
He2
1 1 2 2
± =
H2 Li2: core 1σ Li2: core 1σ∗ Li2: Valence 2σ Li2: Total
O2 molecule Total 2σ and 2σ∗ 3σ and 1π 1π∗
Nature 2004 vol 432 867
1 2 2 2 SPz z
±
Incorrect!