SLIDE 1
Constitutional isomers 1 6 6 Two bracelets with the same - - PowerPoint PPT Presentation
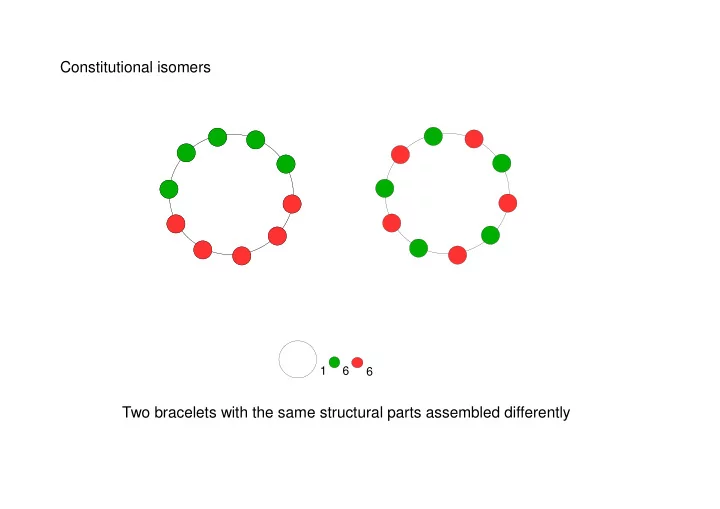
Constitutional isomers 1 6 6 Two bracelets with the same - - PowerPoint PPT Presentation
Constitutional isomers 1 6 6 Two bracelets with the same structural parts assembled differently Constitutional isomers H H H H H H H C H C C H C N H N H H H H C 2 H 7 N Two molecules with the same constituent atoms
SLIDE 2
SLIDE 3
Rules for writing the Lewis structure of a molecule: 1) MF, constitution, and net charge must be known 2) Draw the dot structures of all the atoms of the MF 3) Determine the total number of valence electrons 4) Connect the atoms by 2 electron covalent bonds 5) Maximize the number of covalent bonds (multiple bonds between atoms are OK) 6) Determine formal and net charge 7) Replace bonded dot pairs with line bonds if desired * * * * * 8) If 2 or more structures (resonance structures) can be drawn, the more representative has a) all octets filled b) minimal formal charge c) has formal charge with the least separation d) has formal charge assigned by relative electronegativities
SLIDE 4
.. F Li :F: ..
- Li +
:.. .. F F .. : : .. .. .. F C H H H .. : ..
Bond polarization: a consequence of electronegativity differences Fluorine, F2 Fluoromethane, CH3F Lithium Fluoride, LiF not ∆EN = (4.0 - 4.0) = 0 F-C (4.0 - 2.5) = 1.5 (4.0 - 1.0) = 3.0 nonpolar covalvent polar covalent bond ionic bond bond C-H (2.5 - 2.1) = 0.4 nonpolar covalent bonds
SLIDE 5
Atomic Orbitals
- Quantum mechanics provides a mathematical
description of electron energies and locations
- It’s called a wave equation
- The solution of the wave equation gives a
series of wavefunctions, ψ1 , ψ2 , ψ3 . . .
- The square of the wavefunction is an atomic
- rbital
SLIDE 6
Atomic Orbital Shapes and Phases
- 5 classes of orbitals
- s, p, d, f, g
2s orbital
see http://www.d.umn.edu/~pkiprof/ChemWebV2/AOs/index.html for orbital diagrams
SLIDE 7
2p orbitals 2py 2px 2pz
SLIDE 8
3d orbitals
SLIDE 9
Wikipedia’s entry on atomic orbitals describes how the wave-like nature of electrons qualitatively determines the shape of an orbital: http://en.wikipedia.org/wiki/Atomic_orbital
SLIDE 10
+ =
- r
. . . .
1s e- not between nuclei if populated 1s = antibonding interaction
σ σ*
- =
. .
1s 1s 1s (intersection) e- between nuclei 1s = bonding interaction (exclusion)
H H
energy internuclear distance
104.2 kcal/mol 0.74 Å
SLIDE 11
+ =
node between nuclei = antibonding interaction
- =
1s 1s 1s (constructive inteference) 1s electron density between nuclei = bonding interaction (destructive interference)
SLIDE 12
Can this approach be extended to methane, CH4? If the idea of LCAO is H 1s1 + H 1s1 = H2 σ2 H2 σ* Yes, but the mathematics is very challenging! C 1s2 + C 2s2 + C 2px
1 + C 2py 1 + C 2pz + H 1s1 + H 1s1 + H 1s1 + H 1s1 = ?
SLIDE 13
So, to model how atomic orbitals overlap to provide valance bonds, we will
- choose to combine only 2 orbitals at a time
- invent hybrid atomic orbitals to reconcile LCAO with VSEPR
SLIDE 14
H Be H H B H H Be H . . . H . H:Be:H B H . . . H . . H . H:B:H .. H C H . . . H . . H . H:C:H .. H . H . .. H H C H H H
Using VSEPR to predict molecular geometries Beryllium hydride, BeH2 Borane, BH3 Methane, CH4 180 E 120 E 109.5 E
linear
linear linear linear trigonal planar trigonal planar trigonal planar trigonal planar tetrahedral tetrahedral tetrahedral tetrahedral
SLIDE 15
π π* C 2p C 2p
energy
. . . .
- r
. . . .
SLIDE 16
H C H H H N H H H .. O H H .. ..
VSEPR geometries can be distorted is the electrostatic interactions between paris of valence elctrons are not identical: lp - lp interactions > lp - bonded pair interactions > bonded pair - bonded pair interactions 109.5 E Methane 107E Ammonia 105 E Water
distorted tetrahedral distorted tetrahedral distorted tetrahedral distorted tetrahedral
SLIDE 17
C N H H H O O . . . . . . . . . .
+
- Dipole moments of molecules: net molecular polarity
Draw the Lewis structure for nitromethane, CH3NO2 nitromethane
SLIDE 18
C N H H H O O . .. . . . . . . .
+
- Dipole moments of molecules: net molecular polarity
quantitatively, µ = Q × r visually, dipole moment can be described as a vector with magnitude and direction that is the sum of the bond polarity vectors of a molecule. Usually, it’s OK to ignore the contributions of formally nonpolar covalent bonds that have ∆EN # 0.4: net
SLIDE 19
C N H H H O O . .. . . . . . . .
+
- Accounting for the polarization of every bond with ∆EN … 0 gives a more accurate µ but rarely
changes the gross direction and magnitude: net
SLIDE 20