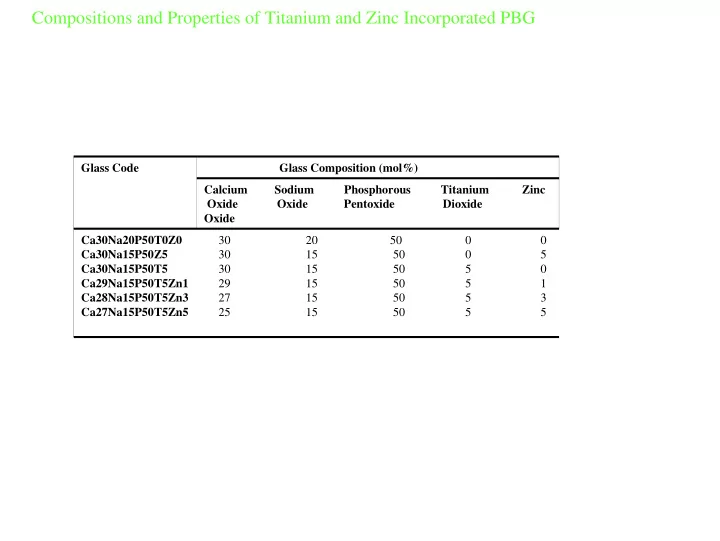
Glass Composition (mol%) Glass Code Calcium Sodium Phosphorous Titanium Zinc Oxide Oxide Pentoxide Dioxide Oxide Ca30Na20P50T0Z0 Ca30Na15P50Z5 Ca30Na15P50T5 Ca29Na15P50T5Zn1 Ca28Na15P50T5Zn3 Ca27Na15P50T5Zn5 30 20 50 0 0 30 15 50 0 5 30 15 50 5 0 29 15 50 5 1 27 15 50 5 3 25 15 50 5 5
Compositions and Properties of Titanium and Zinc Incorporated PBG