Basics of NMR Spectroscopy
N S
Bo B1 Detector
Frequency Generator Recorder
Magnet
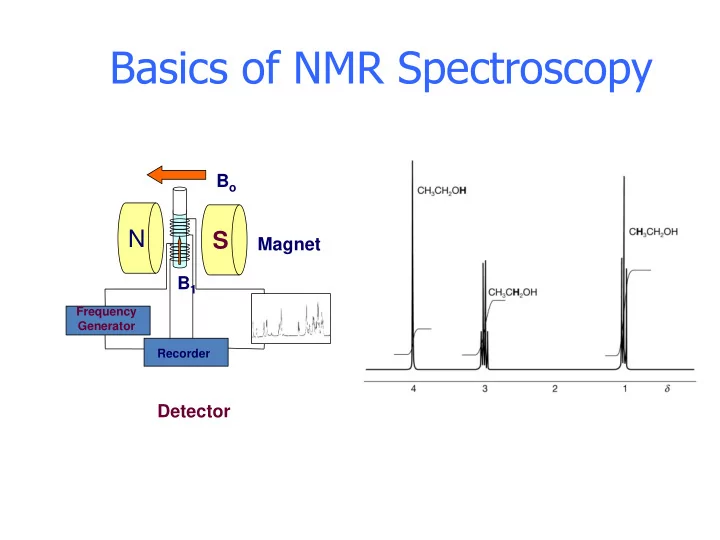
Basics of NMR Spectroscopy B o N S Magnet B 1 Frequency - - PowerPoint PPT Presentation
Basics of NMR Spectroscopy B o N S Magnet B 1 Frequency Generator Recorder Detector Electromagnetic Spectrum Electromagnetic Spectrum Electronic Electromagnetic Spectrum Electronic Vibration Electromagnetic Spectrum Electronic
Bo B1 Detector
Frequency Generator Recorder
Magnet
Electronic
Electronic Vibration
Electronic Vibration Rotation
Electronic Vibration Spin resonance Rotation
Certain nuclei absorb radiofrequencies (electromagnetic radiation) when they are placed in a magnetic field. Essential criterion: spin number (I) 0.
I = 0 (12C, 16O)
I = whole integer (14N, 2H, 10B)
I = half integer (1H, 13C, 15N, 31P) Angular momentum = [I(I+1)]1/2 h/2 Z-component of angular momentum = m h/2 m = I, (I - 1), (I - 2), … , -I For 1H: m = 1/2, -1/2
Certain nuclei absorb radiofrequencies (electromagnetic radiation) when they are placed in a magnetic field. Essential criterion: spin number (I) 0.
I = 0 (12C, 16O)
I = whole integer (14N, 2H, 10B)
I = half integer (1H, 13C, 15N, 31P) Angular momentum = [I(I+1)]1/2 h/2 Z-component of angular momentum = m h/2 m = I, (I - 1), (I - 2), … , -I For 1H: m = 1/2, -1/2
Certain nuclei absorb radiofrequencies (electromagnetic radiation) when they are placed in a magnetic field. Essential criterion: spin number (I) 0.
I = 0 (12C, 16O)
I = whole integer (14N, 2H, 10B)
I = half integer (1H, 13C, 15N, 31P) Angular momentum = [I(I+1)]1/2 h/2 Z-component of angular momentum = m h/2 m = I, (I - 1), (I - 2), … , -I For 1H: m = 1/2, -1/2
Certain nuclei absorb radiofrequencies (electromagnetic radiation) when they are placed in a magnetic field. Essential criterion: spin number (I) 0.
I = 0 (12C, 16O)
I = whole integer (14N, 2H, 10B)
I = half integer (1H, 13C, 15N, 31P) Angular momentum = [I(I+1)]1/2 h/2 Z-component of angular momentum = m h/2 m = I, (I - 1), (I - 2), … , -I For 1H: m = 1/2, -1/2
Certain nuclei absorb radiofrequencies (electromagnetic radiation) when they are placed in a magnetic field. Essential criterion: spin number (I) 0.
I = 0 (12C, 16O)
I = whole integer (14N, 2H, 10B)
I = half integer (1H, 13C, 15N, 31P) Angular momentum = [I(I+1)]1/2 h/2 Z-component of angular momentum = m h/2 m = I, (I - 1), (I - 2), … , -I For 1H: m = 1/2, -1/2
For a steady magnetic field B0, E = -m B0 (m = Magnetic moment = g I ) g = Magnetogyric ratio; għ = gl mN ) mN = eħ/2mp = Nuclear magneton = 5.051 x 10-27 JT-1 gl = Nuclear g factor (Range = -6 to +6), Ĥ = - g B0 î Considering the field to be along the z-direction, mz = gIz = g m ħ; E = - mz B0 = - g m ħ B0 Different spin states have different energies in the presence of a magnetic field
For a steady magnetic field B0, E = -m B0 (m = Magnetic moment = g I ) g = Magnetogyric ratio; għ = gl mN ) mN = eħ/2mp = Nuclear magneton = 5.051 x 10-27 JT-1 gl = Nuclear g factor (Range = -6 to +6), Ĥ = - g B0 î Considering the field to be along the z-direction, mz = gIz = g m ħ; E = - mz B0 = - g m ħ B0 Different spin states have different energies in the presence of a magnetic field
For a steady magnetic field B0, E = -m B0 (m = Magnetic moment = g I ) g = Magnetogyric ratio; għ = gl mN ) mN = eħ/2mp = Nuclear magneton = 5.051 x 10-27 JT-1 gl = Nuclear g factor (Range = -6 to +6), Ĥ = - g B0 î Considering the field to be along the z-direction, mz = gIz = g m ħ; E = - mz B0 = - g m ħ B0 Different spin states have different energies in the presence of a magnetic field
= hL
i.e. L = g B0 / 2
Resonance: The energy of the EM radiation
matches the energy gap B0 = 12T, L = 500 MHz for protons
= hL
i.e. L = g B0 / 2
Resonance: The energy of the EM radiation
matches the energy gap B0 = 12T, L = 500 MHz for protons
= hL
i.e. L = g B0 / 2
L : precessional frequency
The chemical environment alters the effective magnetic field on the nuclei Beff = Bo( 1 - s ) s = magnetic shielding of the nucleus. Factors that affect it include neighboring atoms, aromatic groups, etc., the polarization of the bonds to the observed nuclei
The chemical environment alters the effective magnetic field on the nuclei Beff = Bo( 1 - s ) s = magnetic shielding of the nucleus. Factors that affect it include neighboring atoms, aromatic groups, etc., the polarization of the bonds to the observed nuclei
eff L
The chemical environment alters the effective magnetic field on the nuclei Beff = Bo( 1 - s ) s = magnetic shielding of the nucleus. Factors that affect it include neighboring atoms, aromatic groups, etc., the polarization of the bonds to the observed nuclei
eff L
1H/ 13C nuclei in different environments
resonate at different frequencies
The chemical environment alters the effective magnetic field on the nuclei Beff = Bo( 1 - s ) s = magnetic shielding of the nucleus. Factors that affect it include neighboring atoms, aromatic groups, etc., the polarization of the bonds to the observed nuclei L
low field high field Intensity Population
eff L
1H/ 13C nuclei in different environments
resonate at different frequencies
particular nucleus.
particular nucleus.
particular nucleus.
d is field-independent d (-s)
particular nucleus.
d is field-independent d (-s)
Reference: Tetramethyl silane (TMS) soluble in most organic solvents, inert, volatile, and has 12 equivalent 1Hs and 4 equivalent 13Cs Other references: residual solvent peak, dioxane for 13C, H3PO4 for 31P
H 3 C Si CH3 CH3 CH3
Shielded
Shielded Deshielded
Shielded Deshielded
Shielded Deshielded Shielded
Three groups of lines = Three kinds of protons
Three groups of lines = Three kinds of protons Areas: Relative intensities = Abundance
Three groups of lines = Three kinds of protons Areas: Relative intensities = Abundance
What is the significance
lines?
Br CH3
Small alteration in the magnetic field experienced by a nucleus due to other magnetic nuclei ► Fine structure in the spectra
Br CH3
Small alteration in the magnetic field experienced by a nucleus due to other magnetic nuclei ► Fine structure in the spectra
Br CH3
Small alteration in the magnetic field experienced by a nucleus due to other magnetic nuclei ► Fine structure in the spectra E = J . I1 . I2
Br CH3
Small alteration in the magnetic field experienced by a nucleus due to other magnetic nuclei ► Fine structure in the spectra E = J . I1 . I2 Coupling Constant J
Br CH3
Small alteration in the magnetic field experienced by a nucleus due to other magnetic nuclei ► Fine structure in the spectra E = J . I1 . I2 Interacting spins Coupling Constant J
Br CH3
Small alteration in the magnetic field experienced by a nucleus due to other magnetic nuclei ► Fine structure in the spectra E = J . I1 . I2 Interacting spins Coupling Constant J
Br CH3
Small alteration in the magnetic field experienced by a nucleus due to other magnetic nuclei ► Fine structure in the spectra E = J . I1 . I2 Interacting spins Coupling Constant J