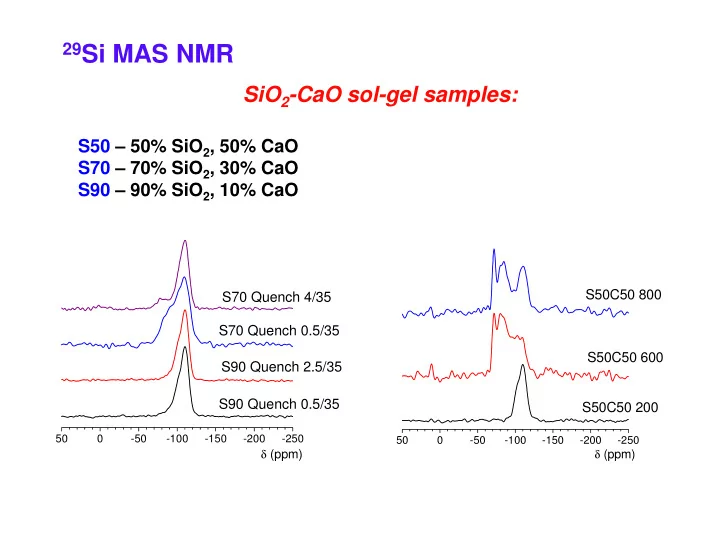
29Si MAS NMR
S50 – 50% SiO2, 50% CaO S70 – 70% SiO2, 30% CaO S90 – 90% SiO2, 10% CaO
SiO2-CaO sol-gel samples:
50
- 50
- 100
- 150
- 200
- 250
S70 Quench 4/35 S70 Quench 0.5/35 S90 Quench 2.5/35 S90 Quench 0.5/35
δ (ppm)
50
- 50
- 100
- 150
- 200
- 250
S50C50 800 S50C50 600 S50C50 200
δ (ppm)