WHATS T-REX? Control the synthesis of any protein of interest - - PowerPoint PPT Presentation
WHATS T-REX? Control the synthesis of any protein of interest - - PowerPoint PPT Presentation
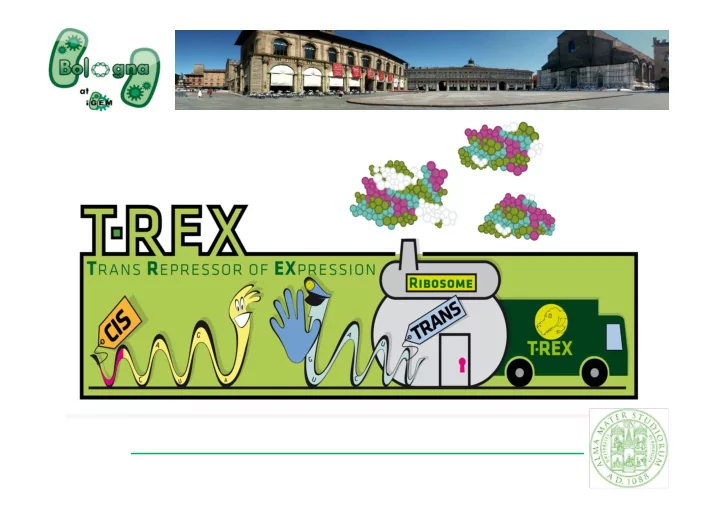
WHATS T-REX? Control the synthesis of any protein of interest Silence the protein expression faster than using classic regulated promoters iGEM2009UniversityofBologna This device is composed of two BioBricks:
WHAT’S T-REX?
- Control the synthesis of any protein of interest
- Silence the protein expression faster than using classic regulated
promoters
iGEM 2009 – University of Bologna
This device is composed of two BioBricks:
- TRANS- repressor
- CIS- repressing
iGEM 2009 – University of Bologna
- Transcription of the target gene yields a mRNA strand;
- The mRNA with the CIS sequence at 5' end, is available for
translation.
iGEM 2009 – University of Bologna
- When the promoter controlling the TRANS coding sequence is active
its transcript binds with the CIS mRNA.
- This RNA duplex prevents ribosomes from binding to RBS, thus
silencing protein synthesis.
iGEM 2009 – University of Bologna
TESTING CIRCUIT
iGEM 2009 – University of Bologna
O2
iGEM 2009 – University of Bologna
1) Maximal free energy in the secondary structure, reducing the probability of its intra‐molecular annealing; 2) Minimal unwanted interac?ons with genomic mRNA; 3) Minimal probability of par?al/shiEed hybridiza?on with complementary strands.
BASER
Best Sequence Research by Andrea and Elisa
iGEM 2009 – University of Bologna
HOW BASER WORKS?
Star?ng from a randomly generated sequence (current sequence); Conformity test: a) more than 5 adjacent repeats of the same nucleo?de; b) restric?on sites; c) RBS sequences;
NO YES
iGEM 2009 – University of Bologna BASER replace 5 nucleto?des randomly (genera?on of new sequence); Evaluate score of new sequence;
Add RBS at 3’ end
Evaluate score of current sequence; Score of new sequence is beSer than score of current sequence?
New sequence is preserved Current sequence is preserved
NO YES
iGEM 2009 – University of Bologna
- BASER calculates a score for the current sequence:
1) the self score; 2) the genomic score; 3) the shifted score; How BASER calculates the score?
iGEM 2009 – University of Bologna
Choose of a CIS sequence AACACAAACTATCACTTTAACAACACATTACATATACATTAAAATATTACAAAGAGGAGAAA (RBS in italic)
iGEM 2009 – University of Bologna
CCTCTTTGTAATATTTTAATGTATATGTAATGTGTTGTTAAAGTGATAGTTTGTGTT with a 7b‐long RBS cover in green Choose of the TRANS sequences CTTTGTAATATTTTAATGTATATGTAATGTGTTGTTAAAGTGATAGTTTGTGTT with a 4b‐long RBS cover in green underlined
iGEM 2009 – University of Bologna
Morphology: ‐ Eccentricity [0,1]; ‐ Area [min,max]; Focus: ‐ Clustering; ‐ High fluorescence; ‐ High cell number; Output: for each bacterium the area in pixels and the fluorescence
VIFluoR
iGEM 2009 – University of Bologna
Par art Char t Characteriza acterization tion
- Promoter Strengths
- Plasmid copy numbers
- Influence of O2 operator
- Interac?on between LacI and O2 operator
pSB3K3 pSB1A2
BBa_J23100 BBa_K07919 BBa_B0034 BBa_J0431 BBa_B0015 BBa_J23100 BBa_B0015 BBa_J23118 BBa_C0012 BBa_B0015
iGEM 2009 – University of Bologna
Pr Promoter Str
- moter Strengths
engths BBa_J23118 BBa_J23118
(1429) (1429)
BBa_K079032 on pSB1A2
BBa_J23100 BBa_B0034
BBa_J04031 BBa_B0015BBa_K079031 on pSB1A2
BBa_J23118
BBa_B0034 BBa_J04031 BBa_B0015vs
BBa_J23100 BBa_J23100
(2547) (2547)
iGEM 2009 – University of Bologna
Pr Promoter Str
- moter Strengths
engths
Methods ‐ DH5α cells ‐ M9 medium ‐ 37° overnight
Imaging Analysis ‐ VIFluoR
- Fluorimeter Analysis
‐ Tecan M200
BBa_J23118 BBa_J23100
iGEM 2009 – University of Bologna
Pr Promoter Str
- moter Strengths
engths
OD/Fluorescence over?me analysis from OD=0.1au ‐ Growth Curve ‐ Fluorescence ‐ Fluorescence/OD ra?o
iGEM 2009 – University of Bologna
Plasmid Cop Plasmid Copy Number y Numbers s pSB1A2 pSB1A2
(high cop (high copy) y)
vs
pSB3K3 pSB3K3
(lo (low/medium cop w/medium copy) y)
BBa_K201003 on pSB1A2
BBa_B0034 BBa_E0040 BBa_B0015 BBa_J23118BBa_K201003 on pSB3K3
BBa_B0034 BBa_E0040 BBa_B0015 BBa_J23118Methods ‐ DH5α cells ‐ M9 medium ‐ 37° overnight
pSB3K3 pSB1A2
Plasmid Cop Plasmid Copy Number y Numbers s
iGEM 2009 – University of Bologna
Imaging Analysis ‐ VIFluoR
- Fluorimeter Analysis
‐ Tecan M200
iGEM 2009 – University of Bologna
Influence of Influence of O2 O2 BBa_K079032 BBa_K079032
(O2 a (O2 absent) bsent)
vs BBa_K201001
BBa_K201001
(O2 pr (O2 present) esent)
BBa_K079032 on pSB1A2
BBa_B0034 BBa_E0040 BBa_B0015 BBa_J23100BBa_K201001 on pSB1A2
BBa_B0034 BBa_E0040 BBa_B0015 BBa_J23100 BBa_K07919Methods ‐ DH5α cell ‐ M9 medium ‐ 37° overnight
iGEM 2009 – University of Bologna
- Fluorimeter Analysis
‐ Victor 2
pSB1A2
Influence of Influence of O2 O2
iGEM 2009 – University of Bologna
Positiv
- sitive Contr
e Control of
- l of T
Testing Cir esting Circuit cuit
BBa_K201002 on pSB3K3
BBa_B0034 BBa_C0012 BBa_B0015 BBa_J23118 BBa_B0034 BBa_B0015 BBa_J23100BBa_K201001 on pSB1A2
BBa_k07919iGEM 2009 – University of Bologna
Imaging Analysis ‐ VIFluoR ‐ several images ‐ >60 bacteria/image
IPT IPTG induction: Sta G induction: Static R tic Response esponse
Methods ‐ DH5α cells ‐ M9 medium ‐ 37° overnight ‐ several IPTG levels
iGEM 2009 – University of Bologna
Fluorimeter Analysis ‐ Tecan M200 ‐ Dilu?on to OD=0.1 ‐ 1° sample: No IPTG ‐ 2° sample: IPTG 100μM
- Growth Curve
- Fluorescence
Methods ‐ DH5α cell ‐ M9 medium ‐ 37° overnight ‐ No IPTG
IPT IPTG induction: Dynamic R G induction: Dynamic Response esponse
iGEM 2009 – University of Bologna
MATHEMATICAL MODEL
- Transcrip?on and transla?on processes were considered similar to a second order
kine?cs, like an enzyma?c reac?on:
iGEM 2009 – University of Bologna
MATHEMATICAL MODEL
iGEM 2009 – University of Bologna
iGEM 2009 – University of Bologna
iGEM 2009 – University of Bologna
PARAMETERS ASSIGNMENT
From Literature
From Experimental Measurement
iGEM 2009 – University of Bologna
PARAMETERS ASSIGNMENT
PROMOTER RATIO =1.2 PLASMID COPY NUMBER RATIO=4.6 We simulated tes?ng circuit when T‐REX device is idle (Ini?al Trans‐DNA = 0)
iGEM 2009 – University of Bologna
LacI SIGMOIDAL REPRESSION CURVE
iGEM 2009 – University of Bologna
We fiSed experimental data in order to iden?fy LacI‐O2 dissocia?on constant and LacI‐IPTG dissocia?on constant
STATIC IPTG INDUCTION
iGEM 2009 – University of Bologna
DYNAMIC IPTG INDUCTION
Fiqng of the 100 µM IPTG dynamic induc?on with ?me‐varying RNA polymerase
iGEM 2009 – University of Bologna
T-REX SIMULATION
iGEM 2009 – University of Bologna
T-REX ST
- REX STOR
ORY
We didn’t manage to get the final circuit because we didn’t achieve the assemblying of the CIS and TRANS parts
Which were the problems?
- 1. Parts are only 100 bp in length: Quan?ty problem, due to purifica?on?
* P1010 death gene liga?on protocol.
- 2. Enzyme efficency is lower with short flanking sequences: Were our diges?ons effec?ve?
* We order longer PCR primers and doubled the diges?on ?me.
CONCLUSIONS
Enter informa?on detailing at least one new standard BioBrick Part or Device in the Registry of Standard Parts and demonstrate that works as expected; Submit DNA for at least one new BioBrick Part or Device to the Registry of Parts.
iGEM 2009 – University of Bologna
CONCLUSIONS
Characterize or improve an exis?ng BioBrick Part or Device and enter this informa?on back on the Registry. Help another iGEM team: Cloned and sent the BioBrick BBa_K201002 to the UNIPV‐Pavia iGEM team.
iGEM 2009 – University of Bologna
HUMAN PRACTICE- SHARING
We realized:
- An Online Survey
- An Information Booklet
iGEM 2009 – University of Bologna
ON-LINE SURVEY RESULTS
- General lack of knowledge about Synthetic Biology
- Most people expressed curiosity about Synthetic Biology and iGEM
- After reading the booklet, great part of the respondent recognized
the importance of a responsible and conscious use of Synthetic Biology
iGEM 2009 – University of Bologna
Total respondents: 484
Acknowledgements
- University of Bologna
- Ser.In.Ar Cesena
- Cultural Association San Sebastiano
Instructors: Silvio Cavalcanti, Francesca Ceroni, Emanuele Domenico Giordano, Alejandro Hochkoeppler, Marco Caprini
iGEM 2009 – University of Bologna
iGEM 2009 – University of Bologna