Unit 3 Day 1.notebook September 24, 2015
Unit 3 Day 1 "Atoms"
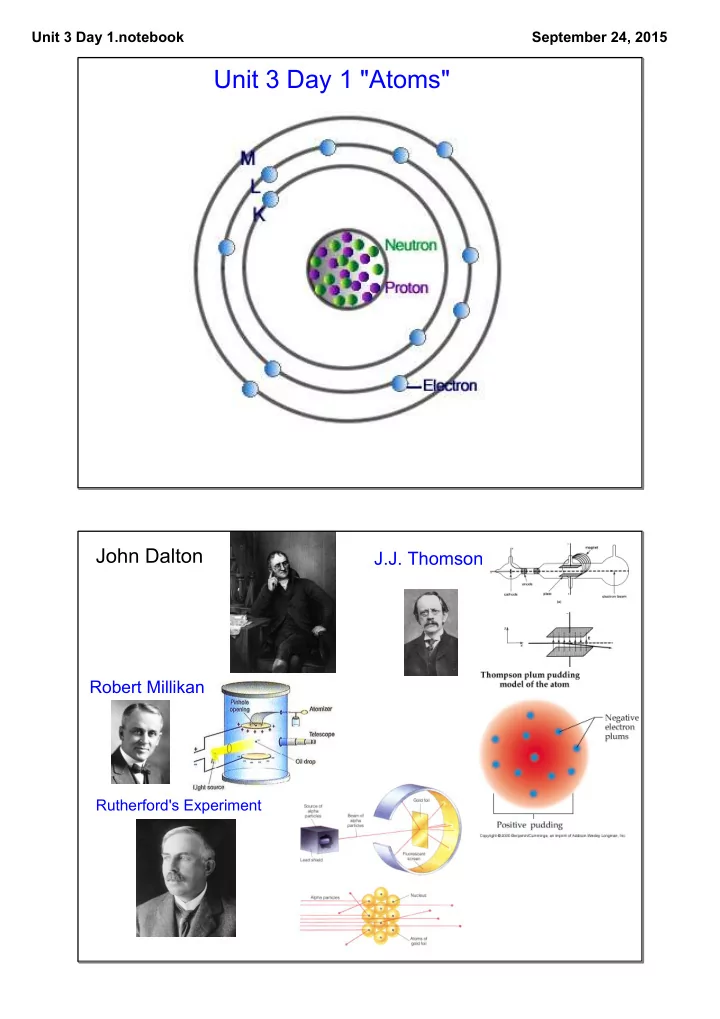
John Dalton
J.J. Thomson
Robert Millikan
Rutherford's Experiment
Unit 3 Day 1 "Atoms" John Dalton J.J. Thomson Robert - - PDF document
Unit 3 Day 1.notebook September 24, 2015 Unit 3 Day 1 "Atoms" John Dalton J.J. Thomson Robert Millikan Rutherford's Experiment Unit 3 Day 1.notebook September 24, 2015 Atomic symbols NOT!!! on a periodic table Mass Number:
Unit 3 Day 1.notebook September 24, 2015
Unit 3 Day 1 "Atoms"
John Dalton
J.J. Thomson
Robert Millikan
Rutherford's Experiment
Unit 3 Day 1.notebook September 24, 2015
symbol Atomic symbols NOT!!! on a periodic table
Atomic Number:
(not an ion) then also the # of e
WHAT IT IS!!!!
neutrons
atomic mass
Charge: only there if it is an ion (lost
electrons)
Said another way:
Bromine81
Unit 3 Day 1.notebook September 24, 2015
symbol
Atomic Number:
Atomic symbols on a periodic table
Average atomic mass:
all the isotopes of the element
100
(27.976927 * 92.23) + (28.976495 * 4.67) + (29.973770 * 3.10) 100
Isotope From Wikipedia, the free encyclopedia Isotopes are variants of a particular chemical element. While all isotopes of a given element share the same number of protons, each isotope differs from the
Calculating Average Atomic Mass
Unit 3 Day 1.notebook September 24, 2015
all the same number (if not an ion) (if an ion add or subtract e
)
# p+ + #n
mass# atomic #
Unit 3 Day 1.notebook September 24, 2015
periods
group number
similar properties
Name of group: Alkali earth metals (other names on pg. 124) Alkali metals Alkaline Earth metals Transition Metals Halogens Nobel or Inert gases
Unit 3 Day 1.notebook September 24, 2015
Half life calculations Nuclear Equations Nuclear Equations
Unit 3 Day 1.notebook September 24, 2015
Half life calculations
Attachments Bohratom.swf Rutherford_s Experiment_ Nuclear Atom1.flv Cathode ray tube and electron.flv