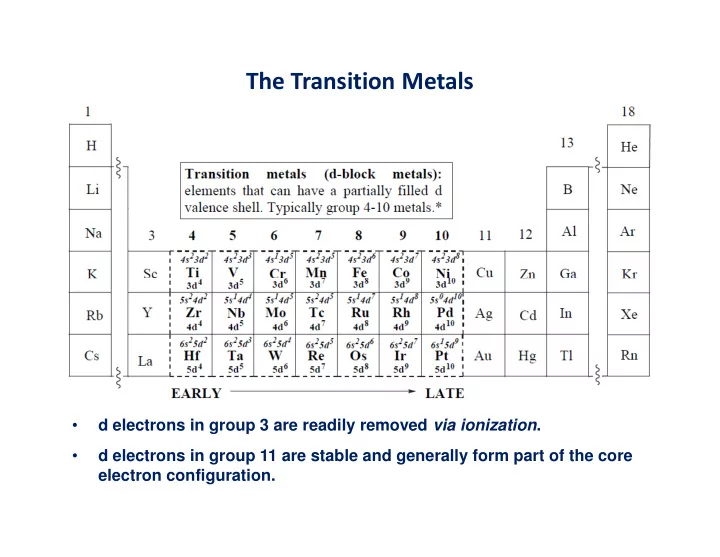
The Transition Metals
- d electrons in group 3 are readily removed via ionization.
- d electrons in group 11 are stable and generally form part of the core
The Transition Metals d electrons in group 3 are readily removed - - PowerPoint PPT Presentation
The Transition Metals d electrons in group 3 are readily removed via ionization . d electrons in group 11 are stable and generally form part of the core electron configuration. Group electronegativity (M) only useful for purely
6
7 Muckerman & Thummel Inorg. Chem. 2008
8
9
10
11
12
13
14
− − −) + [4 x(1 valence e− − − −)] = 8e− − − −
− − −) + [6 x(2 valence e− − − −)] = 18e− − − −
Bridging ligands (multidentate non-chelating)
formal charge # of e−
− − −
donated ionic model
1/metal 2/metal 2/metal 2/metal 1/metal µ µ µ µ-hydride µ µ µ µ-oxo µ µ µ µ-alkoxide µ µ µ µ-halide µ µ µ µ-CO
2/metal 2/metal 2/metal µ µ µ µ-en (ethylene diamine) µ µ µ µ-pyrazine µ µ µ µ-4,4’-bipyridine µ µ µ µ-dppe [1,2-bis(diphenyl phosphino)ethane]
− − − + 2+ =
− − − = 10e− − − −
− − −
− − −
− − −